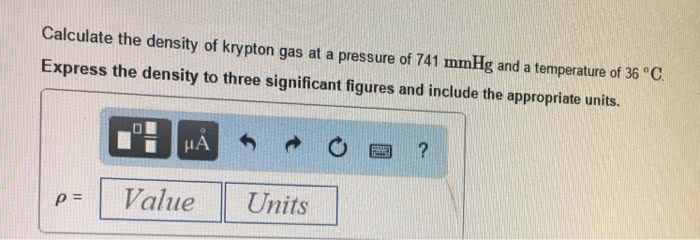
1
answer
0
watching
258
views
11 Dec 2019
Part A) The density of a sample of N2O gas is 3.00 g/L at 298 K.What is the pressure of the gas (in mmHg)?
Part B) An experiment shows that a 110 mL gas sample has a mass of 0.173 g at a pressure of 751 mmHg and a temperature of 30 âC.
What is the molar mass of the gas?
Part C) A gas has a density of 2.07 g/L at a temperature of 24 âC and a pressure of 0.787 atm . Calculate the molar mass of the gas.
Express the molar mass to three significant figures and include the appropriate units.
Part D) Calculate the density of argon gas at a pressure of 749 mmHg and a temperature of 50 âC.
Express the density to three significant figures and include the appropriate units.
Part A) The density of a sample of N2O gas is 3.00 g/L at 298 K.What is the pressure of the gas (in mmHg)?
Part B) An experiment shows that a 110 mL gas sample has a mass of 0.173 g at a pressure of 751 mmHg and a temperature of 30 âC.
What is the molar mass of the gas?
Part C) A gas has a density of 2.07 g/L at a temperature of 24 âC and a pressure of 0.787 atm . Calculate the molar mass of the gas.
Express the molar mass to three significant figures and include the appropriate units.
Part D) Calculate the density of argon gas at a pressure of 749 mmHg and a temperature of 50 âC.
Express the density to three significant figures and include the appropriate units.
Keith LeannonLv2
13 Dec 2019