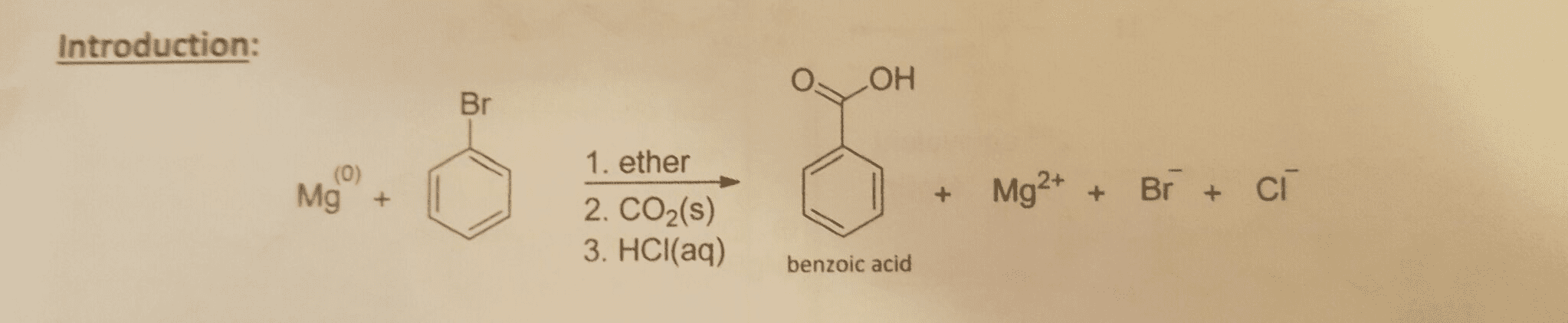
1
answer
0
watching
140
views
11 Dec 2019
In the lab: extraction, purification and analysis of turmeric natural products, Calculate the amount of 3M HCI that would be needed to neutralize the 35 mL of 1M NaOH used in the extractions when you add 20mL 1M NaOH and 10mL saturated sodium chloride to a seperatory funnel which contains 5mL turmeric (extracted from MTBE) and 5 mL of MTBE, shake it, seperating the layers.. You then drain the lower aqueous layer into a flask and leave the top (MTBE) layer in the seperatory funnel and extract it a second time with 15 mL 1M NaOH then combine the NaOH layer with the previous layer in a flask and save for later..
In the lab: extraction, purification and analysis of turmeric natural products, Calculate the amount of 3M HCI that would be needed to neutralize the 35 mL of 1M NaOH used in the extractions when you add 20mL 1M NaOH and 10mL saturated sodium chloride to a seperatory funnel which contains 5mL turmeric (extracted from MTBE) and 5 mL of MTBE, shake it, seperating the layers.. You then drain the lower aqueous layer into a flask and leave the top (MTBE) layer in the seperatory funnel and extract it a second time with 15 mL 1M NaOH then combine the NaOH layer with the previous layer in a flask and save for later..
1
answer
0
watching
140
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232