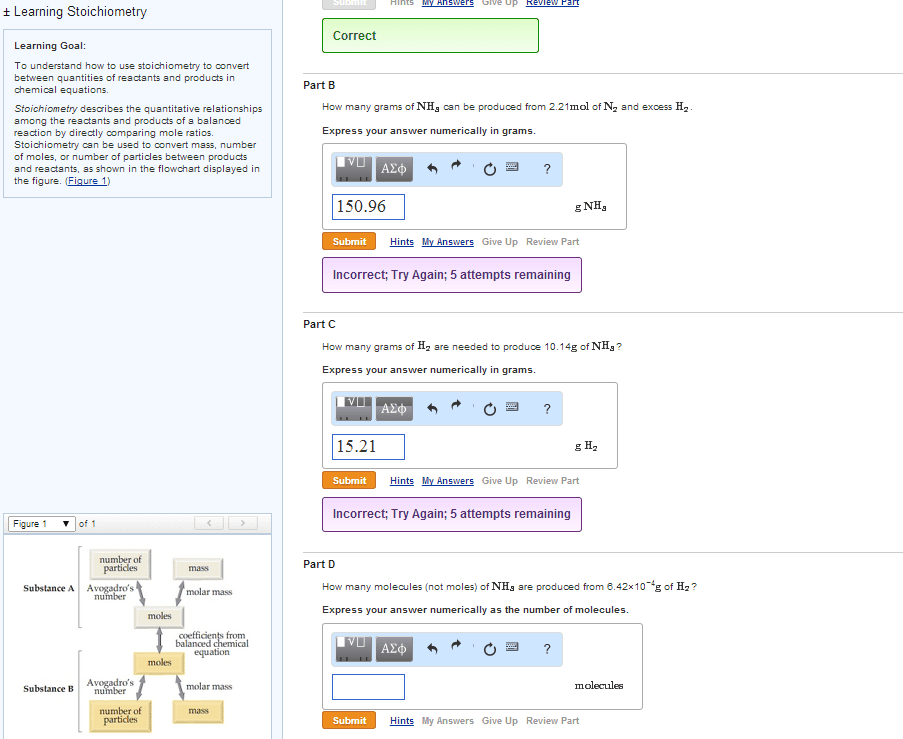
Number Significant figures Explanation
23.56 4 All nonzero digits are significant.
500.01 5 Zeros between nonzero digits are significant.
23.560 5 Trailing zeros are significant when a decimal point isshown.
0.0078 2 Leading zeros are not significant.
1000 ? Trailing zeros at the end of a number but before a decimalare ambiguous.
Part A
Determine the number of significant figures in the measurement 6.07m ?
Express your answer numerically as an integer.
Part B
Determine the number of significant figures in the measurement0.0030 s ?
Express your answer numerically as an integer.
Part C
Round the value 44.981 g to three significant figures.
Express your answer numerically using three significant figures.
Number Significant figures Explanation
23.56 4 All nonzero digits are significant.
500.01 5 Zeros between nonzero digits are significant.
23.560 5 Trailing zeros are significant when a decimal point isshown.
0.0078 2 Leading zeros are not significant.
1000 ? Trailing zeros at the end of a number but before a decimalare ambiguous.
Part A
Determine the number of significant figures in the measurement 6.07m ?
Express your answer numerically as an integer.
Part B
Determine the number of significant figures in the measurement0.0030 s ?
Express your answer numerically as an integer.
Part C
Round the value 44.981 g to three significant figures.
Express your answer numerically using three significant figures.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
| Problem 3.63 Several brands of antacids use Al(OH)3 to react with stomach acid, which contains primarily HCl: αAl(OH)3(s)+βHCl(aq)âγAlCl3(aq)+δH2O(l) | Part A Balance this equation. Express your answer as a balanced chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up Incorrect; Try Again; 5 attempts remaining Part B Calculate the number of grams of HCl that can react with 0.490 g of Al(OH)3.
SubmitMy AnswersGive Up Incorrect; Try Again; 4 attempts remaining Part C Calculate the number of grams of AlCl3 formed when 0.490 g of Al(OH)3 reacts.
SubmitMy AnswersGive Up Incorrect; Try Again; 5 attempts remaining Part D Calculate the number of grams of H2O formed when 0.490 g of Al(OH)3 reacts.
SubmitMy AnswersGive Up Incorrect; Try Again; 4 attempts remaining
|