1
answer
0
watching
109
views
11 Dec 2019
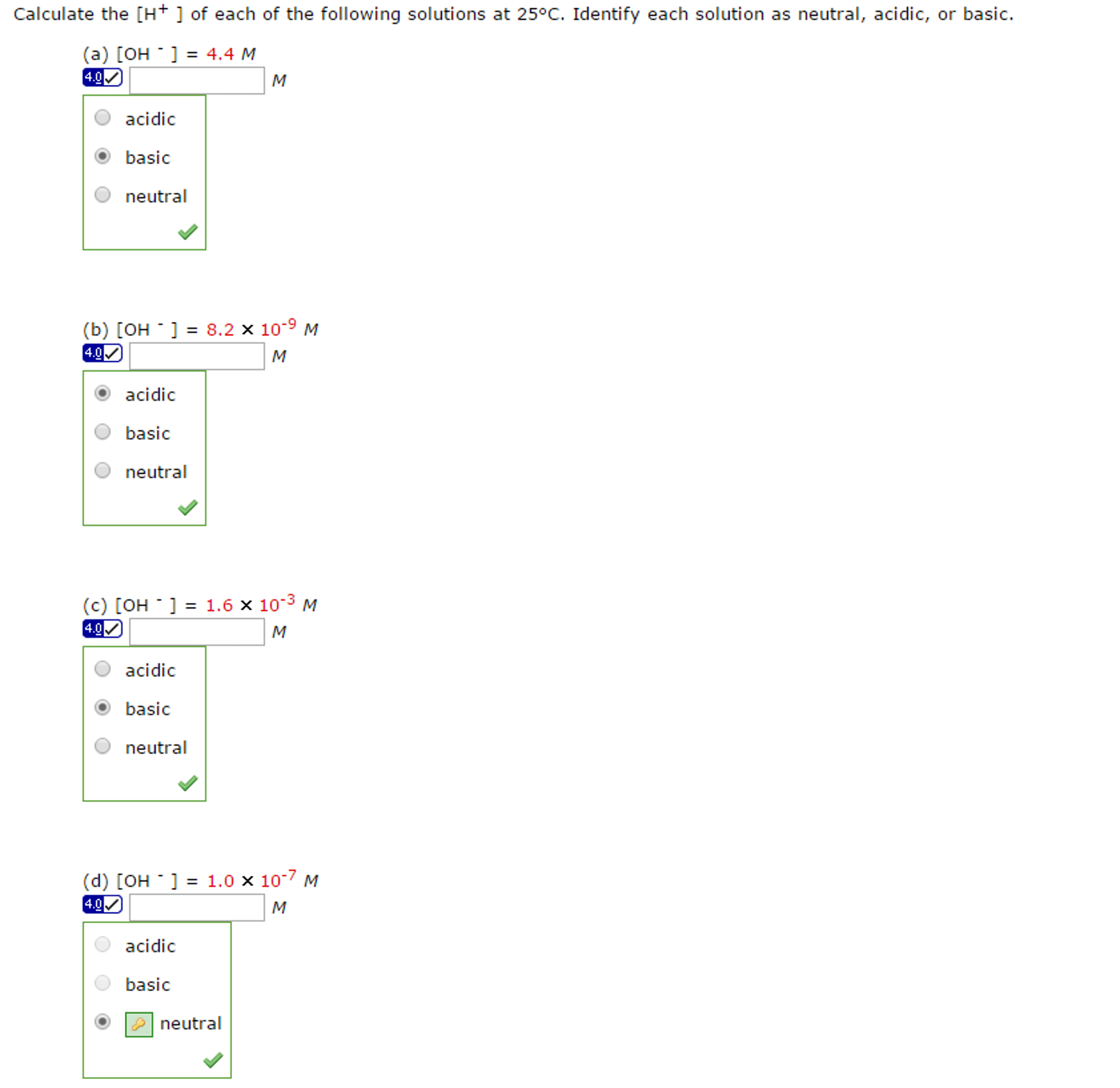
Calculate the pH of each of the following solutions and determine whether the solutions are neutral, acidic, or basic.
a. [H+]= 4.8x10-4 M b. [OH-]=2.5x10-9 M c. [OH-]= 3.2x10-6 M
Calculate the pH of each of the following solutions and determine whether the solutions are neutral, acidic, or basic.
a. [H+]= 4.8x10-4 M b. [OH-]=2.5x10-9 M c. [OH-]= 3.2x10-6 M
Kenneth DuqueLv10
13 Oct 2020