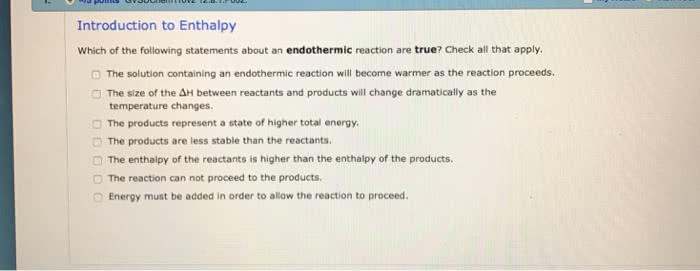
1
answer
1
watching
593
views
grayskunk564Lv1
11 Dec 2019
the overall enthalpy change for a reaction _____.
a. can only be determined by making calorimetric measurements.
b. depends only on the amount of matter that undergoes the change and on the initial state of the reactants and the final state of the products.
c. can only be determined if it is carried out in one step.
d. depends directly on the path by which the reaction is carried out.
the overall enthalpy change for a reaction _____.
a. can only be determined by making calorimetric measurements.
b. depends only on the amount of matter that undergoes the change and on the initial state of the reactants and the final state of the products.
c. can only be determined if it is carried out in one step.
d. depends directly on the path by which the reaction is carried out.
1
answer
1
watching
593
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Iris MandiaLv9
9 Dec 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232