1
answer
1
watching
813
views
lemongnat815Lv1
11 Dec 2019
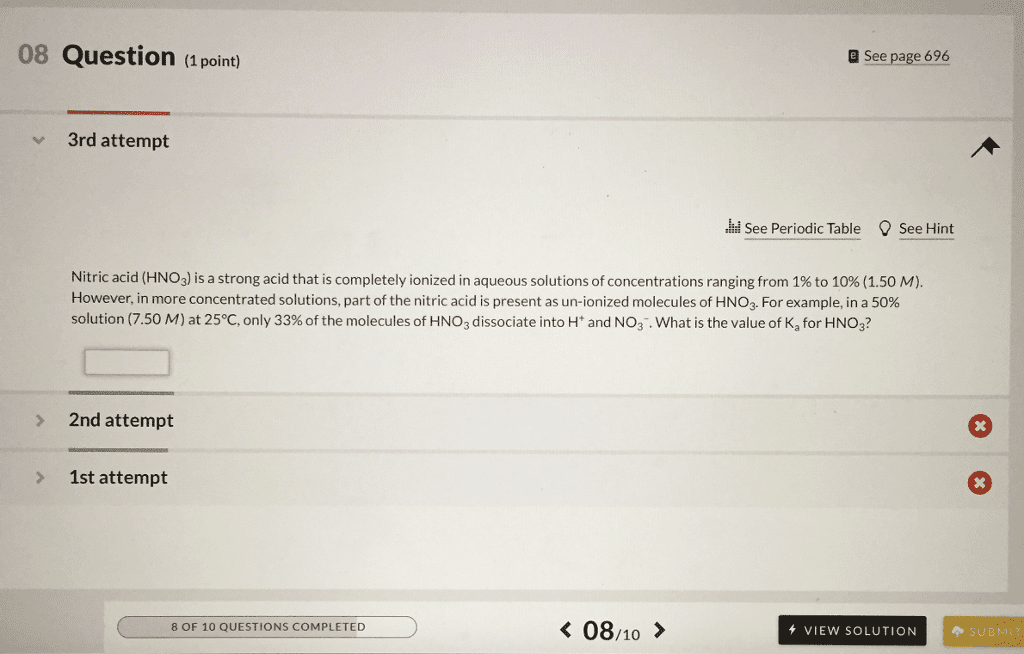
Nitric acid is a strong acid. This means that __________.
a. HNO3 produces a gaseous product when it is neutralized.
b. Aqueous solutions of HNO3 contain equal concentrations of H+(aq) and OH−(aq)
c. HNO3 does not dissociate at all when it is dissolved in water.
d. HNO3 cannot be neutralized by a weak base.
e. HNO3 dissociates completely to H+(aq) and NO3−(aq) when it is dissolved in water.
Nitric acid is a strong acid. This means that __________.
a. HNO3 produces a gaseous product when it is neutralized.
b. Aqueous solutions of HNO3 contain equal concentrations of H+(aq) and OH−(aq)
c. HNO3 does not dissociate at all when it is dissolved in water.
d. HNO3 cannot be neutralized by a weak base.
e. HNO3 dissociates completely to H+(aq) and NO3−(aq) when it is dissolved in water.
1
answer
1
watching
813
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Alyssa Mae RemarimLv10
21 Nov 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232