1
answer
0
watching
229
views
lemoncat98Lv1
11 Dec 2019
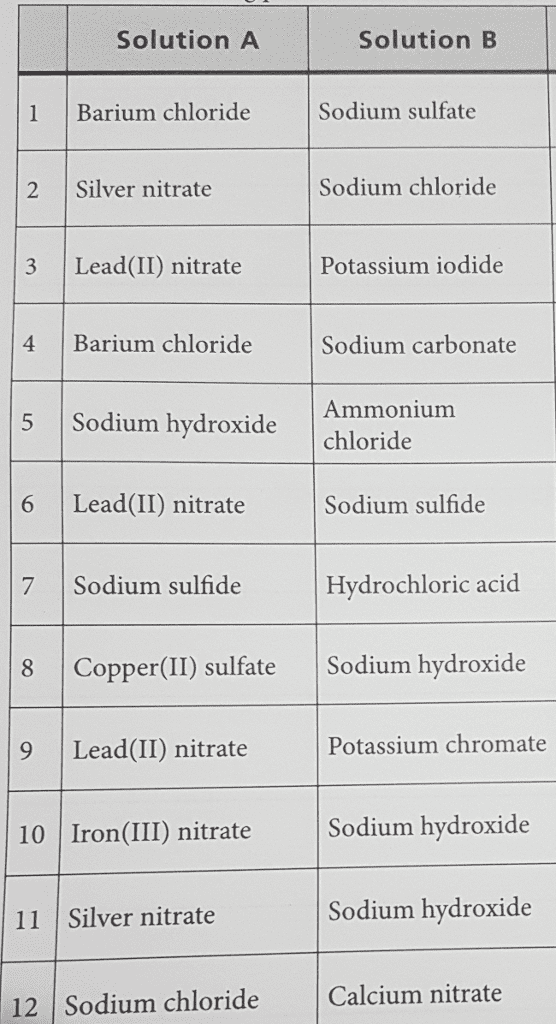
Write a molecular equation for the precipitation reaction that occurs (if any) when the following solutions are mixed. If no reaction occurs, write NO REACTION. Express your answer as a chemical equation. Enter NO REACTION if no reaction occurs. Identify all of the phases in your answer.
A. potassium chloride and lead(II) nitrate
B. lithium nitrate and lead(II) acetate
C. barium nitrate and sodium sulfide
D. strontium nitrate and sodium sulfide
Write a molecular equation for the precipitation reaction that occurs (if any) when the following solutions are mixed. If no reaction occurs, write NO REACTION. Express your answer as a chemical equation. Enter NO REACTION if no reaction occurs. Identify all of the phases in your answer.
A. potassium chloride and lead(II) nitrate
B. lithium nitrate and lead(II) acetate
C. barium nitrate and sodium sulfide
D. strontium nitrate and sodium sulfide
John Edward CayasLv10
28 Oct 2020