1
answer
0
watching
190
views
28 Nov 2019
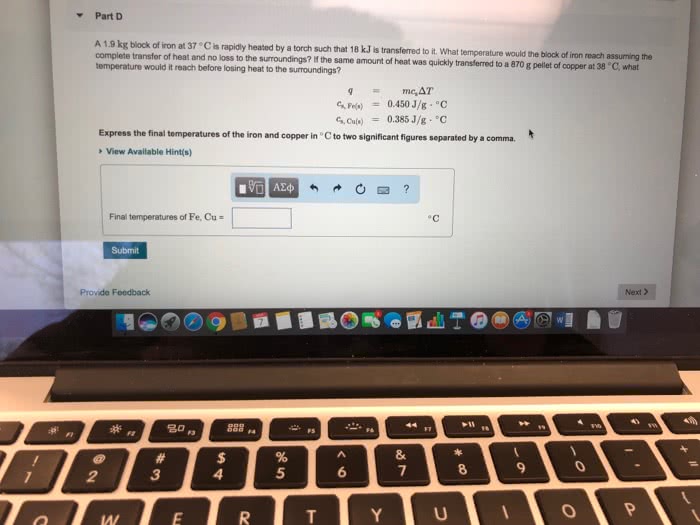
A 1.9 kg block of iron at 23 âC is rapidly heated by a torch such that 17 kJ is transferred to it. What temperature would the block of iron reach assuming the complete transfer of heat and no loss to the surroundings? If the same amount of heat was quickly transferred to a 890 g pellet of copper at 22 âC, what temperature would it reach before losing heat to the surroundings?
Express the final temperatures of the iron and copper in âC to two significant figures separated by a comma.
A 1.9 kg block of iron at 23 âC is rapidly heated by a torch such that 17 kJ is transferred to it. What temperature would the block of iron reach assuming the complete transfer of heat and no loss to the surroundings? If the same amount of heat was quickly transferred to a 890 g pellet of copper at 22 âC, what temperature would it reach before losing heat to the surroundings?
Express the final temperatures of the iron and copper in âC to two significant figures separated by a comma.
Trinidad TremblayLv2
12 Jul 2019