1
answer
0
watching
122
views
27 Nov 2019
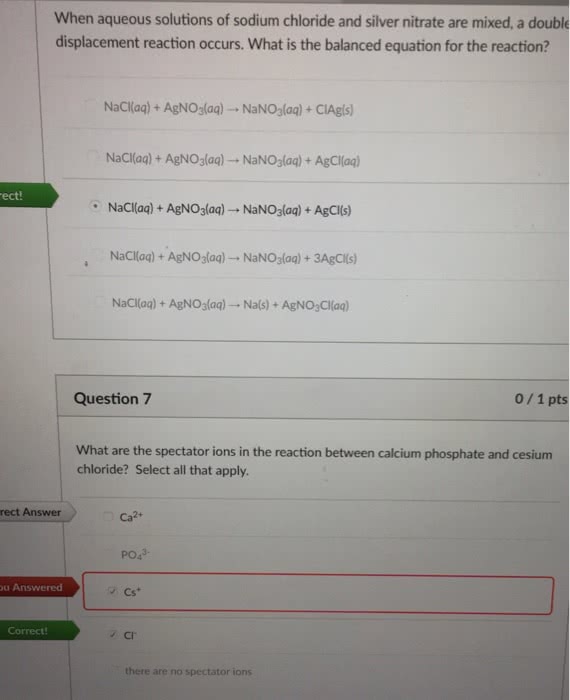
When aqueoussolutions of sodium chloride and silver nitrate are mixed, adouble-displacement reaction occurs. What is thebalanced equation for the reaction? A. NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +AgCl(aq) B. NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +3AgCl(s) C. NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +AgCl(s) D. NaCl(aq) +AgNO3(aq) ? Na(s) +AgNO3Cl(aq) E. NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +ClAg(s)
When aqueoussolutions of sodium chloride and silver nitrate are mixed, adouble-displacement reaction occurs. What is thebalanced equation for the reaction?
| A. | NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +AgCl(aq) | |
| B. | NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +3AgCl(s) | |
| C. | NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +AgCl(s) | |
| D. | NaCl(aq) +AgNO3(aq) ? Na(s) +AgNO3Cl(aq) | |
| E. | NaCl(aq) +AgNO3(aq) ? NaNO3(aq) +ClAg(s) |
Irving HeathcoteLv2
6 May 2019