1
answer
0
watching
353
views
27 Nov 2019
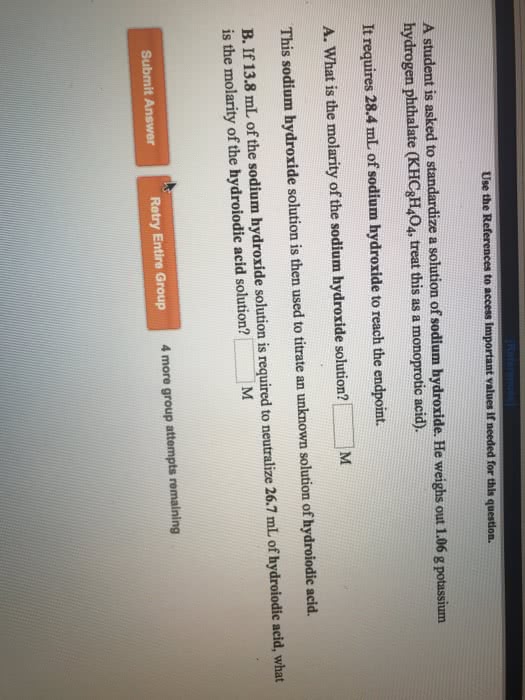
A student is asked to standardize a solution of sodium hydroxide. He weighs out 0.920 g potassium hydrogen phthalate (KHC8H4O4, treat this as a monoprotic acid). It requires 29.5 mL of sodium hydroxide to reach the endpoint.
A.) What is the molarity of the sodium hydroxide solution? 0.153 M This sodium hydroxide solution is then used to titrate an unknown solution of hydroiodic acid.
B.) If 10.7 mL of the sodium hydroxide solution is required to neutralize 25.0 mL of hydroiodic acid, what is the molarity of the hydroiodic acid solution? 0.357 M
A student is asked to standardize a solution of sodium hydroxide. He weighs out 0.920 g potassium hydrogen phthalate (KHC8H4O4, treat this as a monoprotic acid). It requires 29.5 mL of sodium hydroxide to reach the endpoint.
A.) What is the molarity of the sodium hydroxide solution? 0.153 M This sodium hydroxide solution is then used to titrate an unknown solution of hydroiodic acid.
B.) If 10.7 mL of the sodium hydroxide solution is required to neutralize 25.0 mL of hydroiodic acid, what is the molarity of the hydroiodic acid solution? 0.357 M
Jean KeelingLv2
27 Nov 2019