0
answers
0
watching
196
views
27 Nov 2019
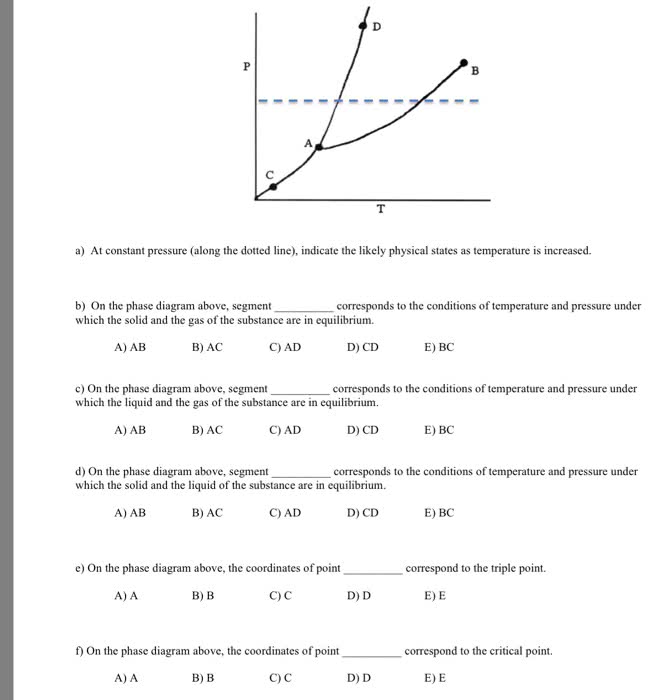
The phase diagram of NH3 can be characterized by the followinginformation. The normal melting and boiling temperatures are 195.2and 239.82 K, respectively, the triple point pressure andtemperature are 6077 Pa and 195.41 K, respectively. The criticalpoint parameters are 112.8 Ã 105 Pa and 405.5 K. Make a sketch ofthe P-T phase diagram (not necessarily to scale) for NH3. Make apoint in the phase diagram for the following conditions. Statewhich and how many phases are present.
a) 195.41 K, 1050 Pa
b) 195.41 K, 6077 Pa
c) 237.51 K, 101325 Pa
d) 420 K, 130 105 Pa
e) 190 K, 6077 Pa
The phase diagram of NH3 can be characterized by the followinginformation. The normal melting and boiling temperatures are 195.2and 239.82 K, respectively, the triple point pressure andtemperature are 6077 Pa and 195.41 K, respectively. The criticalpoint parameters are 112.8 Ã 105 Pa and 405.5 K. Make a sketch ofthe P-T phase diagram (not necessarily to scale) for NH3. Make apoint in the phase diagram for the following conditions. Statewhich and how many phases are present.
a) 195.41 K, 1050 Pa
b) 195.41 K, 6077 Pa
c) 237.51 K, 101325 Pa
d) 420 K, 130 105 Pa
e) 190 K, 6077 Pa