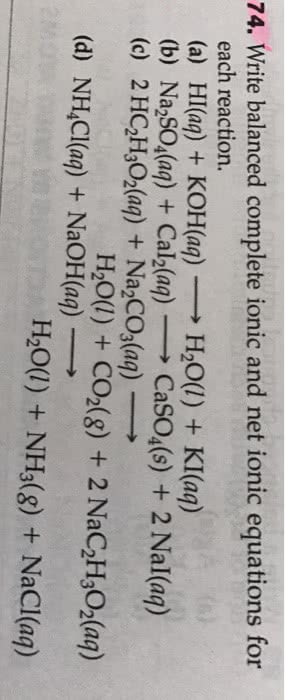1
answer
0
watching
141
views
27 Nov 2019
Predicting Products
Complete and balance the following reactions. Write complete ionicand net ionic equations for all reactions in solution.
1) KOH(aq) + HNO3(aq) ?
2) Na2CO3(aq) + BaCl2(aq) ?
3) Na2SO4(aq) + BaCl2(aq) ?
4) Na3PO4(aq) + CuBr2(aq) ?
5) Ba(OH)2(aq) + HCl(aq) ?
6) NaCl(aq) + K2SO4(aq) ?
7) Ca(OH)2(aq) + HC2H3O2(aq) ?
8) Sr(C2H3O2)2(aq) + NiSO4(aq) ?
9) AgNO3(aq) + Na2CO3(aq) ?
10) Ba(OH)2(aq) + Fe2(SO4)3(aq) ?
11) Cr(OH)3(s) + HNO3(aq) ?
12) Pb(NO3)2(aq) + K2S(aq) ?
13) Fe(OH)3(s) + HClO4(aq) ?
Predicting Products
Complete and balance the following reactions. Write complete ionicand net ionic equations for all reactions in solution.
1) KOH(aq) + HNO3(aq) ?
2) Na2CO3(aq) + BaCl2(aq) ?
3) Na2SO4(aq) + BaCl2(aq) ?
4) Na3PO4(aq) + CuBr2(aq) ?
5) Ba(OH)2(aq) + HCl(aq) ?
6) NaCl(aq) + K2SO4(aq) ?
7) Ca(OH)2(aq) + HC2H3O2(aq) ?
8) Sr(C2H3O2)2(aq) + NiSO4(aq) ?
9) AgNO3(aq) + Na2CO3(aq) ?
10) Ba(OH)2(aq) + Fe2(SO4)3(aq) ?
11) Cr(OH)3(s) + HNO3(aq) ?
12) Pb(NO3)2(aq) + K2S(aq) ?
13) Fe(OH)3(s) + HClO4(aq) ?
Jean KeelingLv2
1 Nov 2019