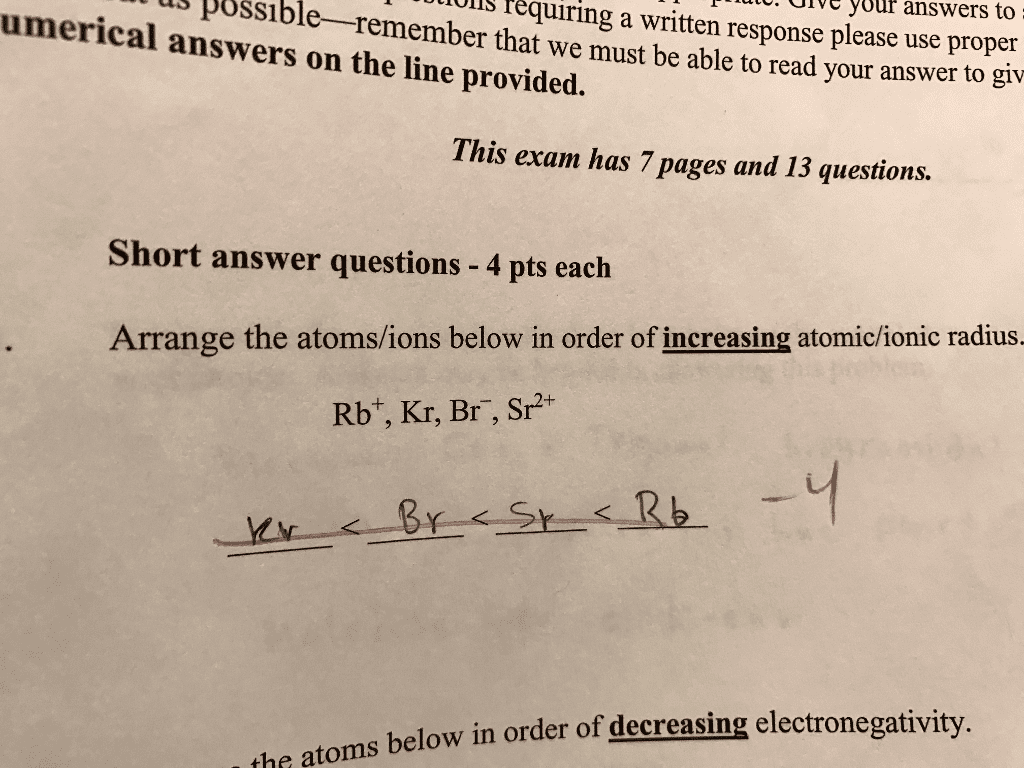2
answers
0
watching
128
views
27 Nov 2019
Arrange the bonds in order of increasingpolarity:
C=N, C-N, C=O,B-O, B-S, B-F
Sol:
Electronegativity difference: C-P (0.4), N-F (1.0),H-O (1.4), B-S (0.5), Si-Cl (1.2)
Order: C-P < B-S < N-F < Si-Cl < H-O
*BUT EXPLAIN HOW??? I DIDNT UNDERSTAND IT
Arrange the bonds in order of increasingpolarity:
C=N, C-N, C=O,B-O, B-S, B-F
Sol:
Electronegativity difference: C-P (0.4), N-F (1.0),H-O (1.4), B-S (0.5), Si-Cl (1.2)
Order: C-P < B-S < N-F < Si-Cl < H-O
*BUT EXPLAIN HOW??? I DIDNT UNDERSTAND IT
25 Apr 2023
Beverley SmithLv2
11 Feb 2019
Already have an account? Log in