0
answers
0
watching
194
views
27 Nov 2019
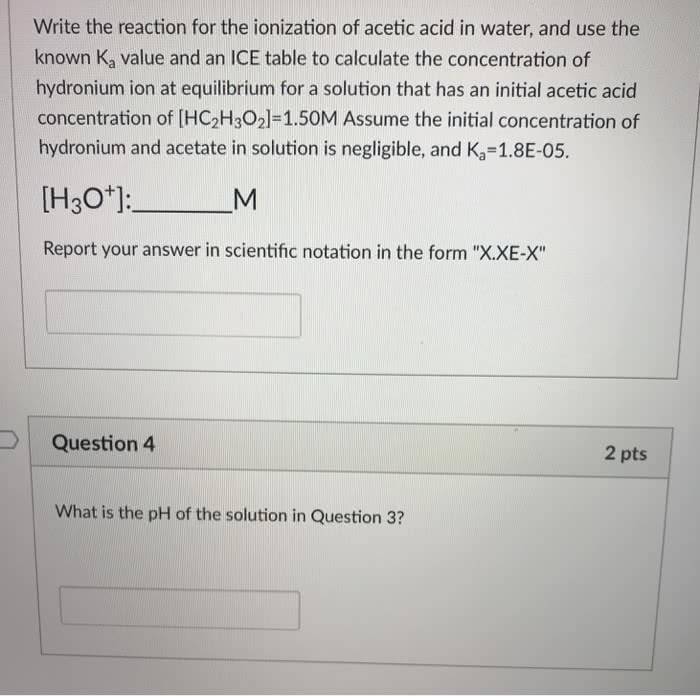
The active ingredient in vinegar is acetic acid, HCH3CO2. It reactswith water to
produce the hydronium ion,
HCH3CO2(aq) + H2O(l) ?? CH3CO2-(aq) + H3O+(aq)
(H+(aq) is a shorthand notation for the hydronium ion). A solutionof acetic acid is prepared
with an initial concentration of acetic acid of 0.100 M. After thesolution goes to equilibrium,
the concentration of the hydronium ion is found to be 1.34 x 10-3 Mat 25.0
The active ingredient in vinegar is acetic acid, HCH3CO2. It reactswith water to
produce the hydronium ion,
HCH3CO2(aq) + H2O(l) ?? CH3CO2-(aq) + H3O+(aq)
(H+(aq) is a shorthand notation for the hydronium ion). A solutionof acetic acid is prepared
with an initial concentration of acetic acid of 0.100 M. After thesolution goes to equilibrium,
the concentration of the hydronium ion is found to be 1.34 x 10-3 Mat 25.0