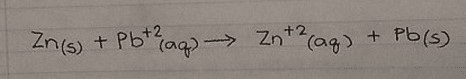0
answers
0
watching
192
views
27 Nov 2019
complete the half reactions for the cell shown here and show theshorthand notation for the cell by dragging labels to the correctposition. The electrode on the left is the anode, and the one onthe right is the cathode.
in the left side there is a solution of Pbcl2(s) and it containsthe elctrode pb and on the right side there is solution of AgCl(S)which contains the electrode Ag. There is inverted glass tube withKcl (aq) on the left and and Kcl(aq) on the right.
Anode half-reaction:
Blank + 2Cl^- equilibrium arrow yields Blank + 2e^-
Cathode half reaction:
Blank +2e^- equlibriums arrow yields Blank + 2Cl^-
Shorthand notation: Please provide.
complete the half reactions for the cell shown here and show theshorthand notation for the cell by dragging labels to the correctposition. The electrode on the left is the anode, and the one onthe right is the cathode.
in the left side there is a solution of Pbcl2(s) and it containsthe elctrode pb and on the right side there is solution of AgCl(S)which contains the electrode Ag. There is inverted glass tube withKcl (aq) on the left and and Kcl(aq) on the right.
Anode half-reaction:
Blank + 2Cl^- equilibrium arrow yields Blank + 2e^-
Cathode half reaction:
Blank +2e^- equlibriums arrow yields Blank + 2Cl^-
Shorthand notation: Please provide.