0
answers
0
watching
118
views
27 Nov 2019
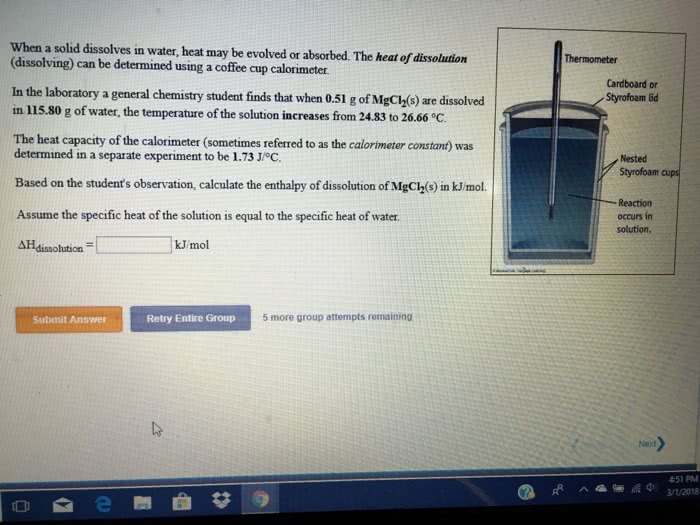
In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used to determine the specific heat of a solid, or to measure the energy of a solution phase reaction. A chunk of copper weighing 19.97 grams and originally at 97.97oC is dropped into an insulated cup containing 79.56 grams of water at 21.22oC. The heat capacity of the calorimeter (sometimes referred to as the calorimeter constant) was determined in a separate experiment to be 1.55 J/oC.Using the accepted value for the specific heat of copper (Appendix link on left), calculate the final temperature of the water. Assume that no heat is lost to the surroundings
In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used to determine the specific heat of a solid, or to measure the energy of a solution phase reaction. A chunk of copper weighing 19.97 grams and originally at 97.97oC is dropped into an insulated cup containing 79.56 grams of water at 21.22oC. The heat capacity of the calorimeter (sometimes referred to as the calorimeter constant) was determined in a separate experiment to be 1.55 J/oC.Using the accepted value for the specific heat of copper (Appendix link on left), calculate the final temperature of the water. Assume that no heat is lost to the surroundings