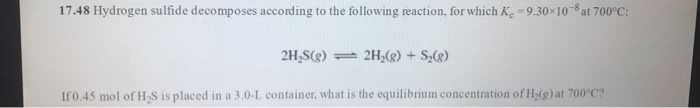1
answer
0
watching
179
views
27 Nov 2019
Hydrogen sulfide decomposes according to the following reaction forwhich Kc=9.3*10^-8 at 700 degrees celcius2H2S(g)<--->2H2(g)+S2(g) if 0.41 mol of H2S is placed in a 3L container, what is the equilibrium eoncentration of H2(g) at 700degrees celcius
Hydrogen sulfide decomposes according to the following reaction forwhich Kc=9.3*10^-8 at 700 degrees celcius2H2S(g)<--->2H2(g)+S2(g) if 0.41 mol of H2S is placed in a 3L container, what is the equilibrium eoncentration of H2(g) at 700degrees celcius
Tod ThielLv2
24 Jun 2019