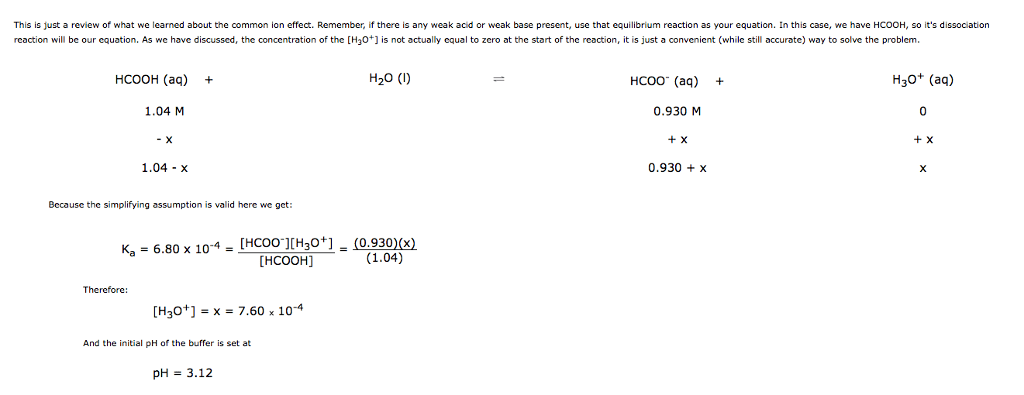1
answer
1
watching
292
views
26 Nov 2019
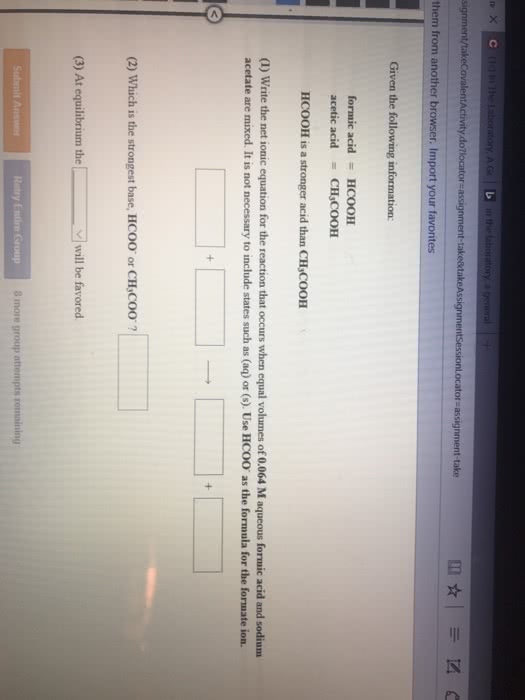
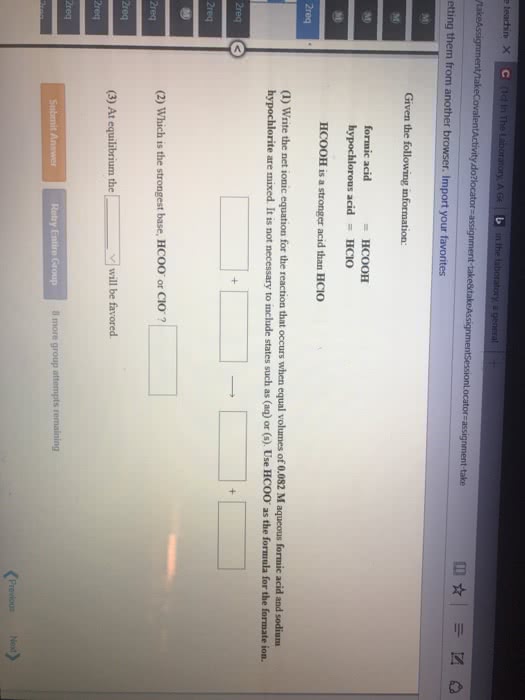
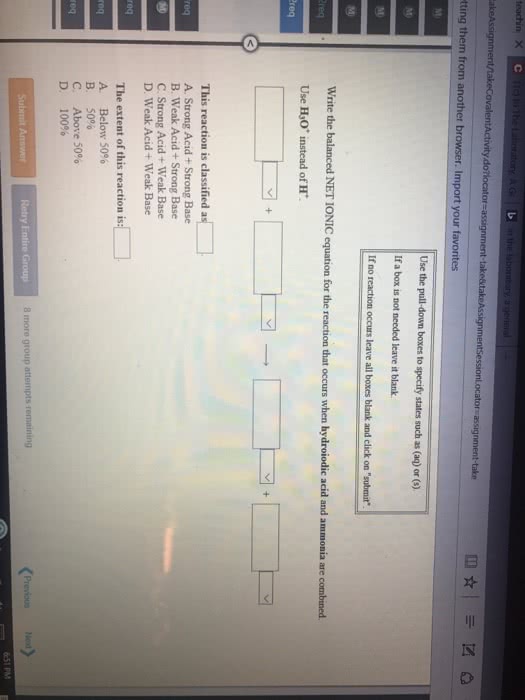
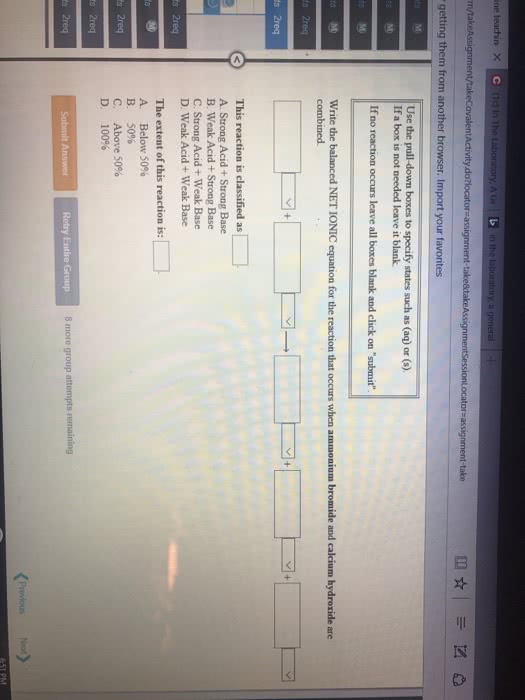
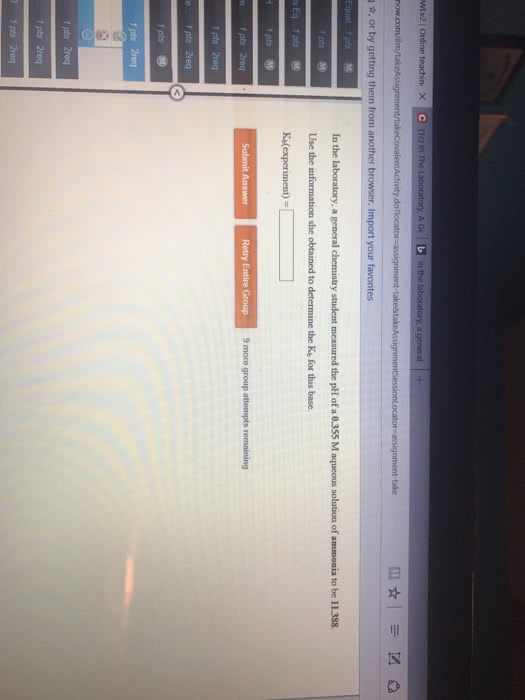
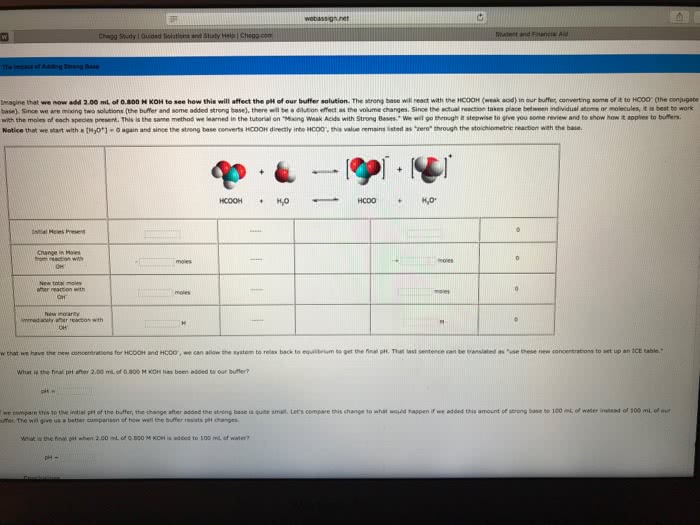
Consider the reaction HCOOH(aq) + Br2(aq) --> 2H+(aq) + 2Br-(aq) + CO2(aq). This reaction is known to be first order in Br2 and zeroth order in HCOOH with k=0.0866 s-1. Write the rate law for this reaction. What is the rate of this reaction when [Br2]=0.500 M and [HCOOH]=0.250 M?
Consider the reaction HCOOH(aq) + Br2(aq) --> 2H+(aq) + 2Br-(aq) + CO2(aq). This reaction is known to be first order in Br2 and zeroth order in HCOOH with k=0.0866 s-1. Write the rate law for this reaction. What is the rate of this reaction when [Br2]=0.500 M and [HCOOH]=0.250 M?
Jarrod RobelLv2
13 Jun 2019