0
answers
0
watching
199
views
26 Nov 2019
30. Suppose two systems, each composed of three particlesrepresented by circles, have 30J of total energy. How manyenergetically equivalent ways can you distribute the particles ineach system? Which system has greater entropy?
System A
10J ooo
System B
12J o
10J o
8J o
30. Suppose two systems, each composed of three particlesrepresented by circles, have 30J of total energy. How manyenergetically equivalent ways can you distribute the particles ineach system? Which system has greater entropy?
System A
10J ooo
System B
12J o
10J o
8J o
0
answers
0
watching
199
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Consider the two diagrams showing the energies (boxes) of each of four A particles and four B particles shown below. The dotted lines represent the allowed energies of each particle.
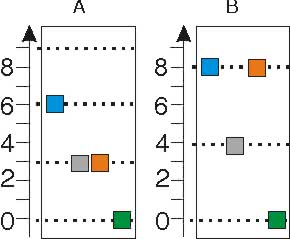 | What is the energy of system A? | 12 |
| What is the energy of system B? | 20 | |
| In how many ways can the energy of system A be distributed? | ? | |
| In how many ways can the energy of system B be distributed? | ? | |
| Which system has the greater entropy? | A |