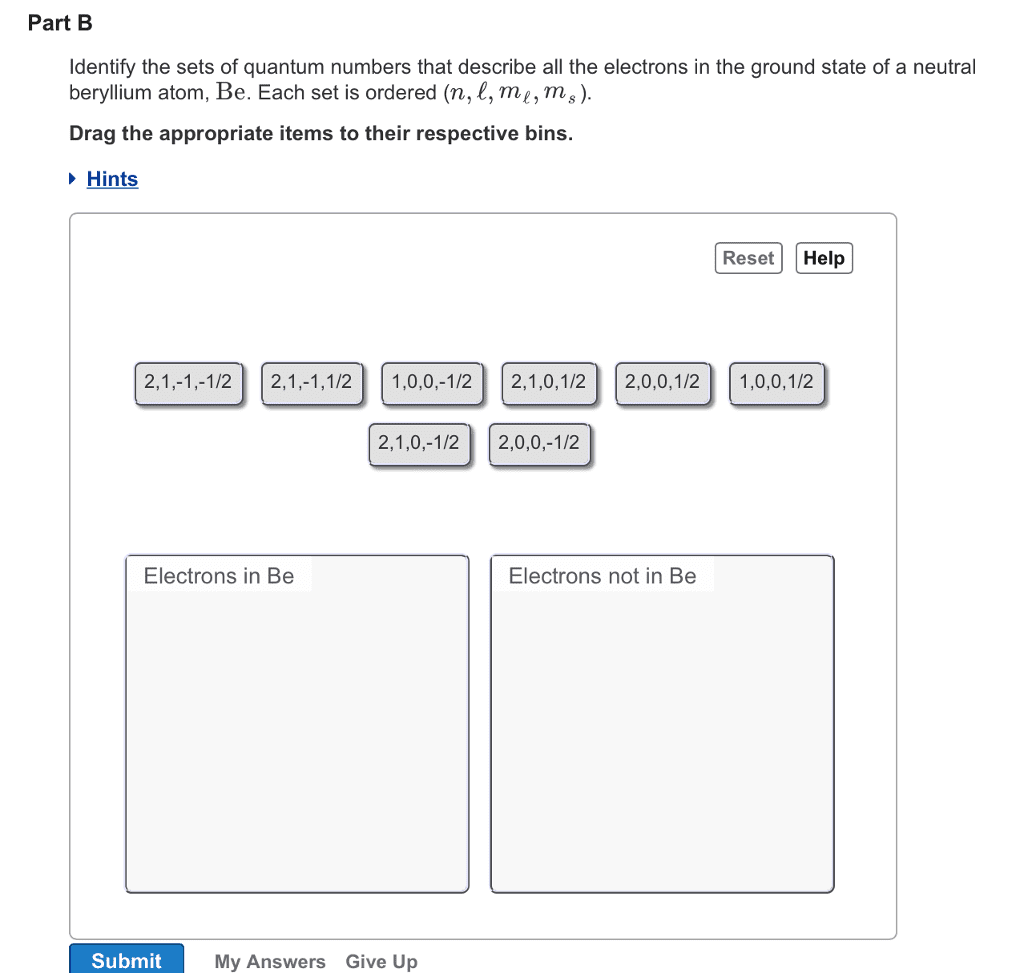
1
answer
0
watching
1,669
views
23 Nov 2019
1)What is the maximum number of electrons in an atom that can have the following quantum numbers?
Part A
n = 3, ml = -2
Express your answer as an integer. Number of electrons =
Part B
n = 4, l = 3
Express your answer as an integer. Number of electrons =
Part C
n = 5, l = 3, ml = 2
Express your answer as an integer. Number of electrons =
Part D
n = 4, l = 1, ml = 0
Express your answer as an integer. Number of electrons =
2)Which of the following set of quantum numbers (ordered n, â, mâ, ms) are possible for an electron in an atom?
Check all that apply.
2, 4, 1, -1/2 3, 2, 2, -1/2 5, 2, 1, -1/2 3, 2, 0, -1/2 4, 3, -4, -1/2 -4, 3, 1, 1/2 2, 1, 3, 1/2 2, 1, 0, -1
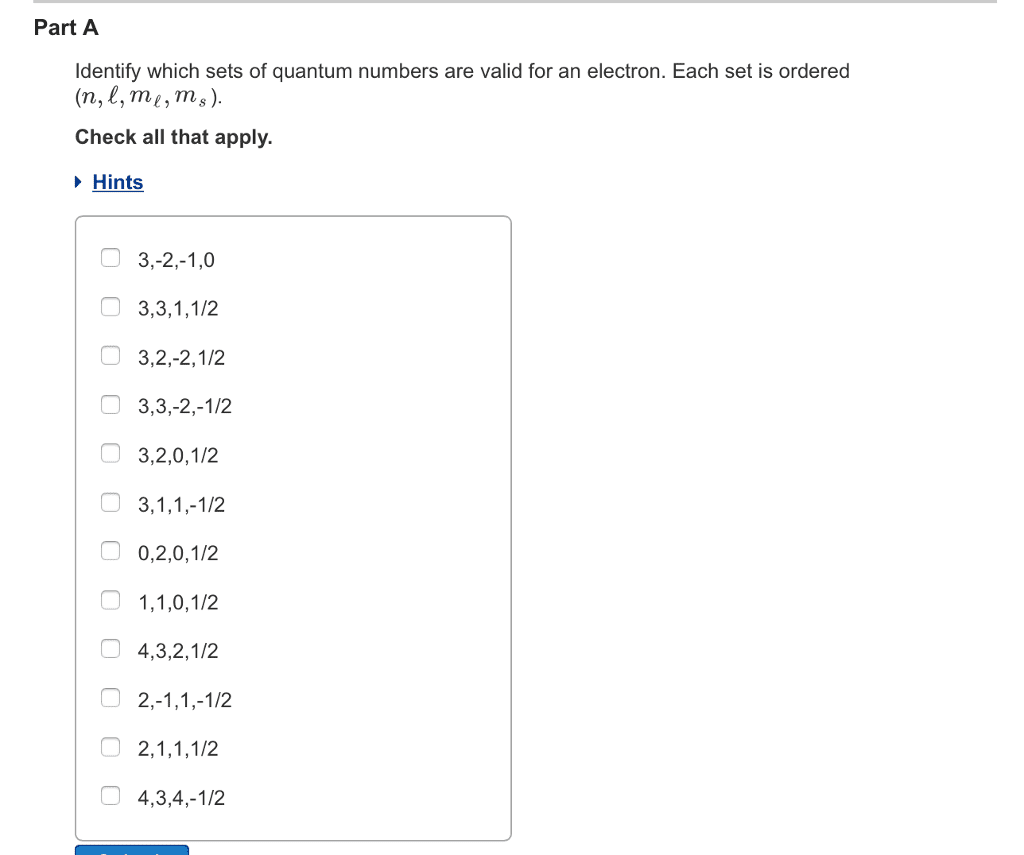
3)Identify which sets of quantum numbers are valid for an electron. Each set is ordered (n,â,mâ,ms).
Check all that apply.
3,2,2,1/2 4,3,2,1/2 1,0,0,-1/2 1,3.0,1/2 3,1,-1,1/2 3,3,-2,-1/2 4,3,4,-1/2 1,1,0,-1/2 0,2,0,1/2 2,1,-1,-1/2 2,-1,1,-1/2 3,2,1,1
1)What is the maximum number of electrons in an atom that can have the following quantum numbers?
Part A
n = 3, ml = -2
Express your answer as an integer. Number of electrons =
Part B
n = 4, l = 3
Express your answer as an integer. Number of electrons =
Part C
n = 5, l = 3, ml = 2
Express your answer as an integer. Number of electrons =
Part D
n = 4, l = 1, ml = 0
Express your answer as an integer. Number of electrons =
2)Which of the following set of quantum numbers (ordered n, â, mâ, ms) are possible for an electron in an atom?
Check all that apply.
| 2, 4, 1, -1/2 | |
| 3, 2, 2, -1/2 | |
| 5, 2, 1, -1/2 | |
| 3, 2, 0, -1/2 | |
| 4, 3, -4, -1/2 | |
| -4, 3, 1, 1/2 | |
| 2, 1, 3, 1/2 | |
| 2, 1, 0, -1 |
3)Identify which sets of quantum numbers are valid for an electron. Each set is ordered (n,â,mâ,ms).
Check all that apply.
| 3,2,2,1/2 | |
| 4,3,2,1/2 | |
| 1,0,0,-1/2 | |
| 1,3.0,1/2 | |
| 3,1,-1,1/2 | |
| 3,3,-2,-1/2 | |
| 4,3,4,-1/2 | |
| 1,1,0,-1/2 | |
| 0,2,0,1/2 | |
| 2,1,-1,-1/2 | |
| 2,-1,1,-1/2 | |
| 3,2,1,1 |
Keith LeannonLv2
6 May 2019