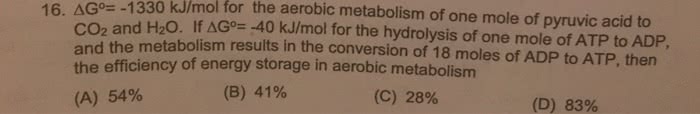0
answers
0
watching
86
views
23 Nov 2019
A major source of energy in aerobic organisms is thecombustionof glucose: C6H12O6 +6O2---> 6CO2 + 6H2O ÎHo' =-2816kJ/mol ÎSo'= +181 J/mol a) at 37oC, what is the value ofÎGo' forthis reaction? b) if during the metabolism of 1 mole of glucose, 38 molesofATP are produced from ADP, and the ÎGo' forthehydrolysis of ATP to ADP is -31 kJ/mol, what is theoverallÎGo' for the combustion of glucose coupled toATPformation? c) What is the efficiency of the process in terms ofthepercentage of available free energy captured in ATP?
A major source of energy in aerobic organisms is thecombustionof glucose:
C6H12O6 +6O2---> 6CO2 + 6H2O
ÎHo' =-2816kJ/mol ÎSo'= +181 J/mol
a) at 37oC, what is the value ofÎGo' forthis reaction?
b) if during the metabolism of 1 mole of glucose, 38 molesofATP are produced from ADP, and the ÎGo' forthehydrolysis of ATP to ADP is -31 kJ/mol, what is theoverallÎGo' for the combustion of glucose coupled toATPformation?
c) What is the efficiency of the process in terms ofthepercentage of available free energy captured in ATP?