1
answer
0
watching
176
views
23 Nov 2019
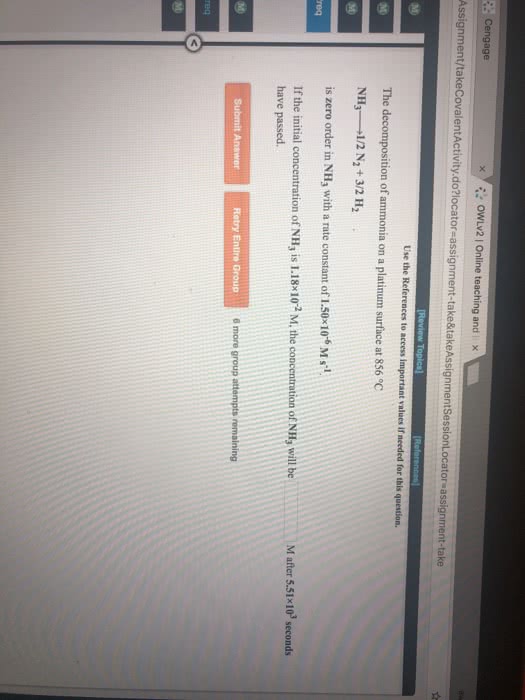
The decomposition of ammonia on a metal surface is a zero order reaction. What is the half-life of the reaction if the initial concentration of ammonia was 0.033 M and 0.015 after 450.0 seconds?
The decomposition of ammonia on a metal surface is a zero order reaction. What is the half-life of the reaction if the initial concentration of ammonia was 0.033 M and 0.015 after 450.0 seconds?
Tod ThielLv2
7 Oct 2019