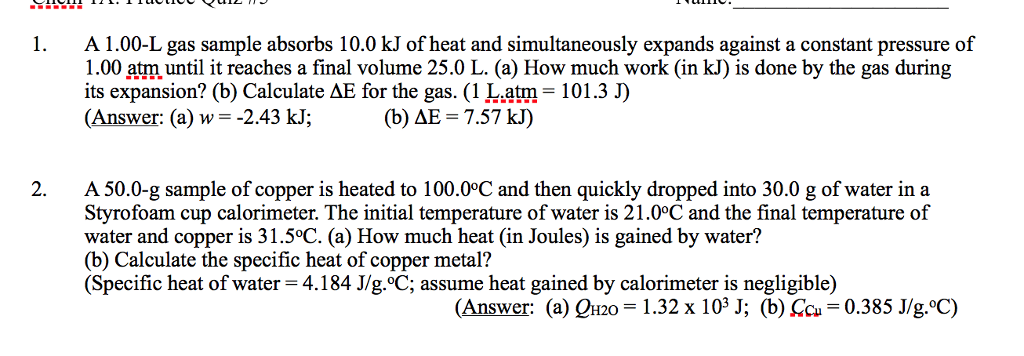1
answer
1
watching
1,595
views
23 Nov 2019
3. Calculate q when 28.6 g of water is heated from 22.0?C to78.3?C.
(the specific heat capacity for water = 4.184 J/(gâ¢oC).
A. 0.385 kJ B. 1.61 kJ C. 6.74 kJ D. 9.37 kJ E. 1.61 x 10^3kJ
help with formula and steps will get you life saver!
3. Calculate q when 28.6 g of water is heated from 22.0?C to78.3?C.
(the specific heat capacity for water = 4.184 J/(gâ¢oC).
A. 0.385 kJ B. 1.61 kJ C. 6.74 kJ D. 9.37 kJ E. 1.61 x 10^3kJ
help with formula and steps will get you life saver!
Reid WolffLv2
28 Aug 2019