2
answers
0
watching
816
views
29 Sep 2019
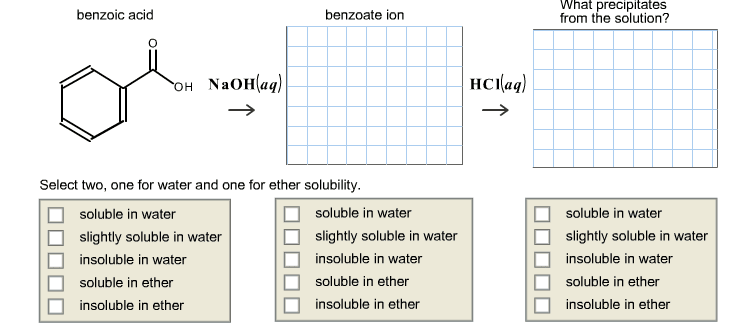
Cyclohexanecarboxylic acid, C6H11COOH (pKa 4.90), is only slightly soluble in water, but its sodium salt,C6H11COO-Na+, is quite soluble in water. Describe the solubility of cyclohexanecarboxylic acid in solutions of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3). The pKa values for the conjugate acids of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3) are 15.7, 6.36, and 10.33, respectively.
Aqueous NaOH: _________soluble, insoluble, or slightly soluble
Aqueous NaHCO3: _________soluble, insoluble, or slightly soluble
Aqueous Na2CO3: _________soluble, insoluble, or slightly soluble
Cyclohexanecarboxylic acid, C6H11COOH (pKa 4.90), is only slightly soluble in water, but its sodium salt,C6H11COO-Na+, is quite soluble in water. Describe the solubility of cyclohexanecarboxylic acid in solutions of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3). The pKa values for the conjugate acids of sodium hydroxide, sodium bicarbonate (NaHCO3), and sodium carbonate (Na2CO3) are 15.7, 6.36, and 10.33, respectively.
Aqueous NaOH: _________soluble, insoluble, or slightly soluble
Aqueous NaHCO3: _________soluble, insoluble, or slightly soluble
Aqueous Na2CO3: _________soluble, insoluble, or slightly soluble
Trinidad TremblayLv2
29 Sep 2019
Already have an account? Log in