1
answer
0
watching
128
views
23 Nov 2019
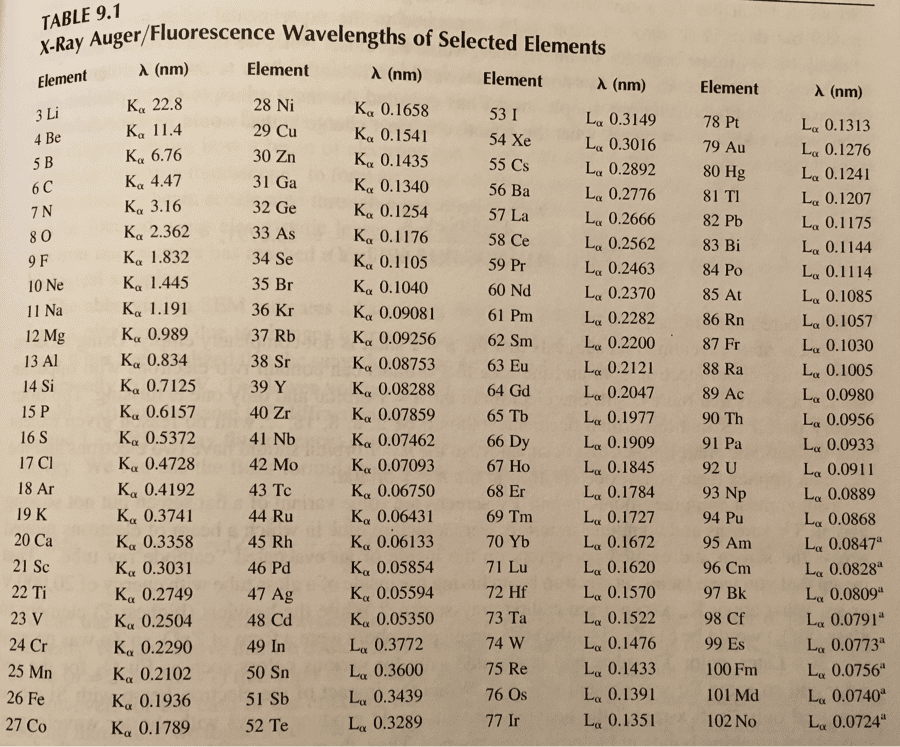
a. 35 b. 45 c. 46 d.70 e. 81. The answer is 46. But I got 47 in my calculation somehow. Since Mg is 2+, I looked at all the elements that has a 2-, which are O, S, Se, Te, Po. Since the question said that its mass is 81, i chose Se since it's the closest to it. Se has atomic number of 34, meaning it has 34 protons. Mass number is from proton and neutron. So I subtracted 81-34: 47. Why is it 46??
a. 35 b. 45 c. 46 d.70 e. 81. The answer is 46. But I got 47 in my calculation somehow. Since Mg is 2+, I looked at all the elements that has a 2-, which are O, S, Se, Te, Po. Since the question said that its mass is 81, i chose Se since it's the closest to it. Se has atomic number of 34, meaning it has 34 protons. Mass number is from proton and neutron. So I subtracted 81-34: 47. Why is it 46??
Irving HeathcoteLv2
7 Jun 2019