1
answer
0
watching
1,132
views
19 Nov 2019
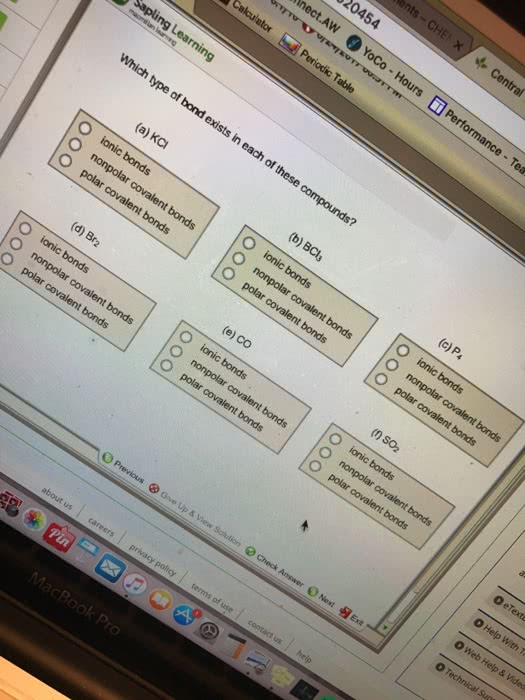

Which type of bond exists in each of these compounds? (a) KCI ionic bonds nonpolar covalent bonds polar covalent bonds (b) BCI_3 ionic bonds nonpolar covalent bonds polar covalent bonds (c) P_4 ionic bonds nonpolar covalent bonds polar covalent bonds (d) Br_2 ionic bonds nonpolar covalent bonds polar covalent bonds (e) CO ionic bonds nonpolar covalent bonds polar covalent bonds (f) ionic bonds nonpolar covalent bonds polar covalent bonds
Hubert KochLv2
5 Jan 2019
