1
answer
0
watching
281
views
19 Nov 2019
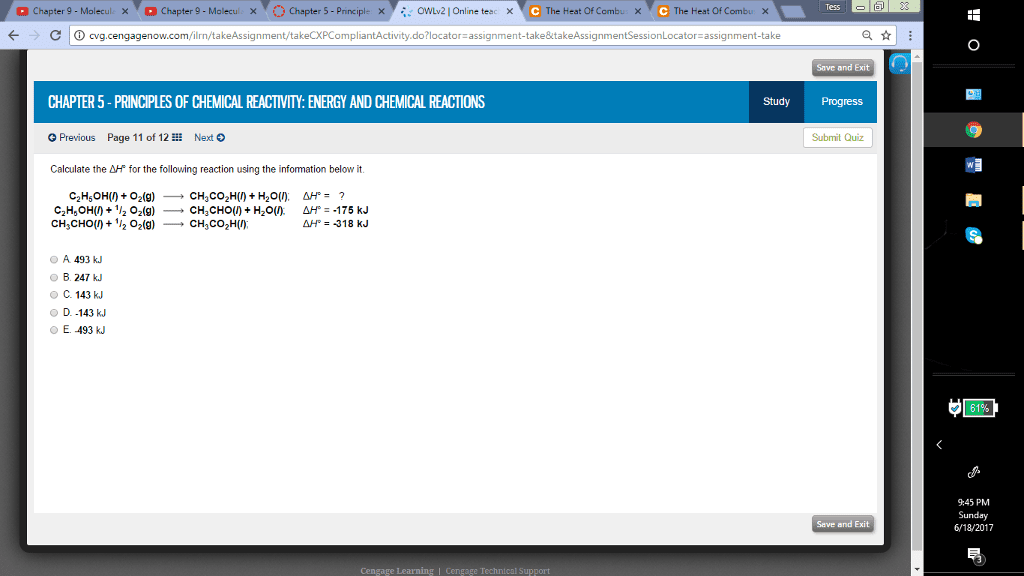
Calculate the delta H^+ for the following reaction using the information below it. C_2H_5OH(l) + O_2 (g) rightarrow CH_3CO_2H(l) + H_2O(l): delta H degree = ? C_2H_5OH_(l) + 1/2 O_2(g) rightarrow CH_3CHO(l) + H_2O(l): delta H degree = -175 kJ CH_3CHO(l) + 1/2 O_2(g) rightarrow CH_3CO_2H(l): delta H degree = -318 kJ 493 kJ 247 kJ 143 kJ -143 kJ -493 kJ

Calculate the delta H^+ for the following reaction using the information below it. C_2H_5OH(l) + O_2 (g) rightarrow CH_3CO_2H(l) + H_2O(l): delta H degree = ? C_2H_5OH_(l) + 1/2 O_2(g) rightarrow CH_3CHO(l) + H_2O(l): delta H degree = -175 kJ CH_3CHO(l) + 1/2 O_2(g) rightarrow CH_3CO_2H(l): delta H degree = -318 kJ 493 kJ 247 kJ 143 kJ -143 kJ -493 kJ
Lelia LubowitzLv2
12 Jul 2019
