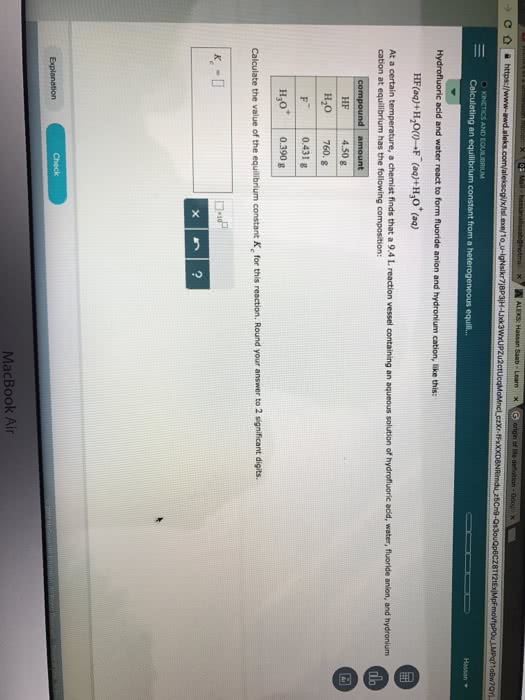
1
answer
0
watching
256
views
18 Nov 2019
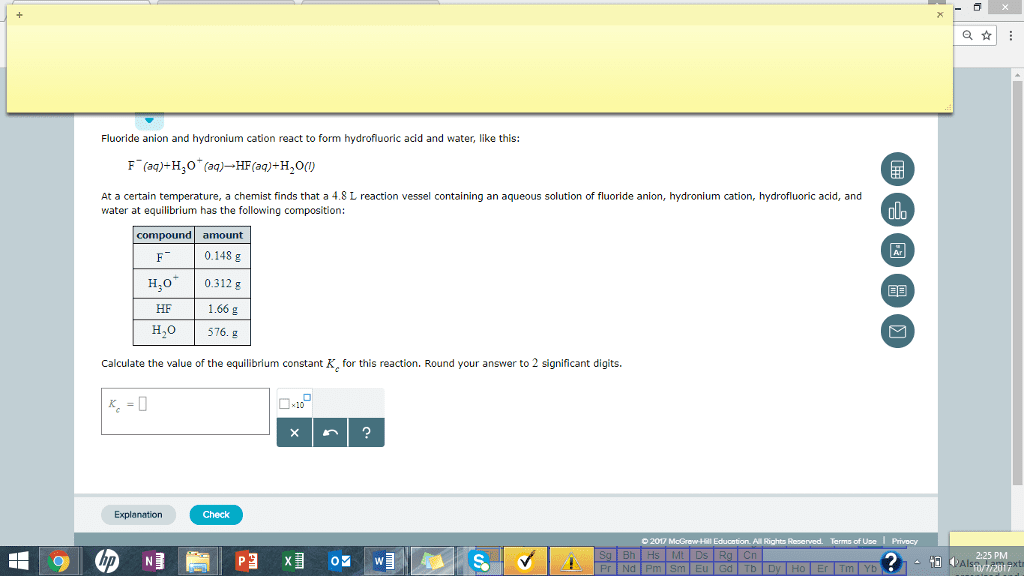
Fluoride anion and hydronium cation react to form hydrofluoric acid and water, like this: At a certain temperature, a chemist finds that a 4.8 L reaction vessel containing an aqueous solution of fluoride anion, hydronium cation, hydrofluoric acid, and water at equilibrium has the following composition: compound amount 0.148 g HO 0.312 g 1.66 g 576. g HF H,o Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. Check © 2017 McGrawHI Edu ation. All Rights Reserved. arm of Use I Privacy 2:25 PM Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb8

Fluoride anion and hydronium cation react to form hydrofluoric acid and water, like this: At a certain temperature, a chemist finds that a 4.8 L reaction vessel containing an aqueous solution of fluoride anion, hydronium cation, hydrofluoric acid, and water at equilibrium has the following composition: compound amount 0.148 g HO 0.312 g 1.66 g 576. g HF H,o Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. Check © 2017 McGrawHI Edu ation. All Rights Reserved. arm of Use I Privacy 2:25 PM Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb8
Nelly StrackeLv2
18 Jun 2019