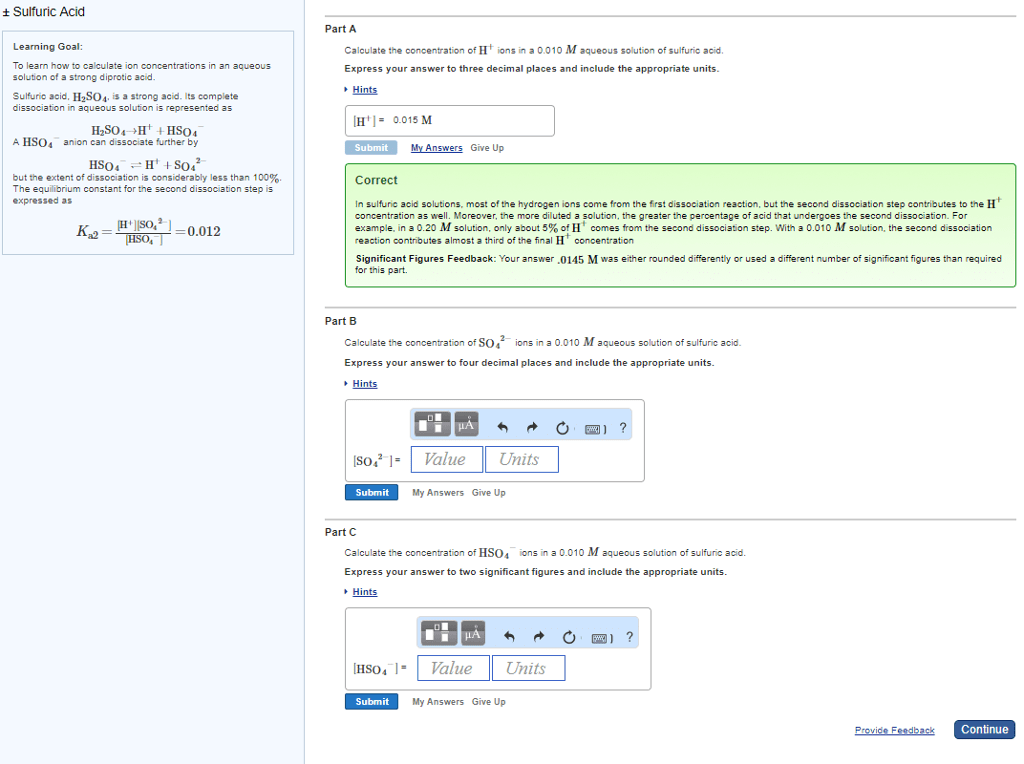
To learn how to calculate ion concentrations in an aqueous solution of a strong diprotic acid.
Sulfuric acid, H2SO4, is a strong acid. Its complete dissociation in aqueous solution is represented as
H2SO4âH++HSO4â
A HSO4â anion can dissociate further by
HSO4ââH++SO42â
but the extent of dissociation is considerably less than 100%. The equilibrium constant for the second dissociation step is expressed as
Ka2=[H+][SO42â][HSO4â]=0.012
Part A
Calculate the concentration of H+ ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to three decimal places and include the appropriate units.
Hints
[H+] =
SubmitMy AnswersGive Up
Part B
Calculate the concentration of SO42â ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to four decimal places and include the appropriate units.
Hints
[SO42â] =
SubmitMy AnswersGive Up
Part C
Calculate the concentration of HSO4â ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to two significant figures and include the appropriate units.
Hints
[HSO4â] =
SubmitMy AnswersGive Up
To learn how to calculate ion concentrations in an aqueous solution of a strong diprotic acid.
Sulfuric acid, H2SO4, is a strong acid. Its complete dissociation in aqueous solution is represented as
H2SO4âH++HSO4â
A HSO4â anion can dissociate further by
HSO4ââH++SO42â
but the extent of dissociation is considerably less than 100%. The equilibrium constant for the second dissociation step is expressed as
Ka2=[H+][SO42â][HSO4â]=0.012
Part A
Calculate the concentration of H+ ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to three decimal places and include the appropriate units.
Hints
| | |||
| [H+] = |
SubmitMy AnswersGive Up
Part B
Calculate the concentration of SO42â ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to four decimal places and include the appropriate units.
Hints
| | |||
| [SO42â] = |
SubmitMy AnswersGive Up
Part C
Calculate the concentration of HSO4â ions in a 0.010 M aqueous solution of sulfuric acid.
Express your answer to two significant figures and include the appropriate units.
Hints
| | |||
| [HSO4â] = |
SubmitMy AnswersGive Up