1
answer
0
watching
1,015
views
18 Nov 2019
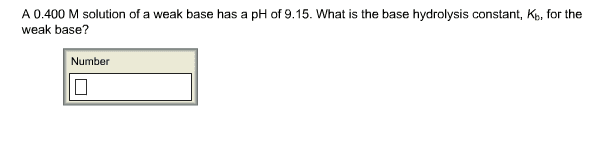
A 0.400 M solution of a weak base has a pH of 9.15. What is the base hydrolysis constant, Kb, for the weak base? Number
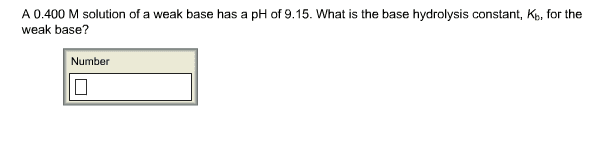
A 0.400 M solution of a weak base has a pH of 9.15. What is the base hydrolysis constant, Kb, for the weak base? Number
Jarrod RobelLv2
19 Jul 2019



