1
answer
0
watching
133
views
18 Nov 2019
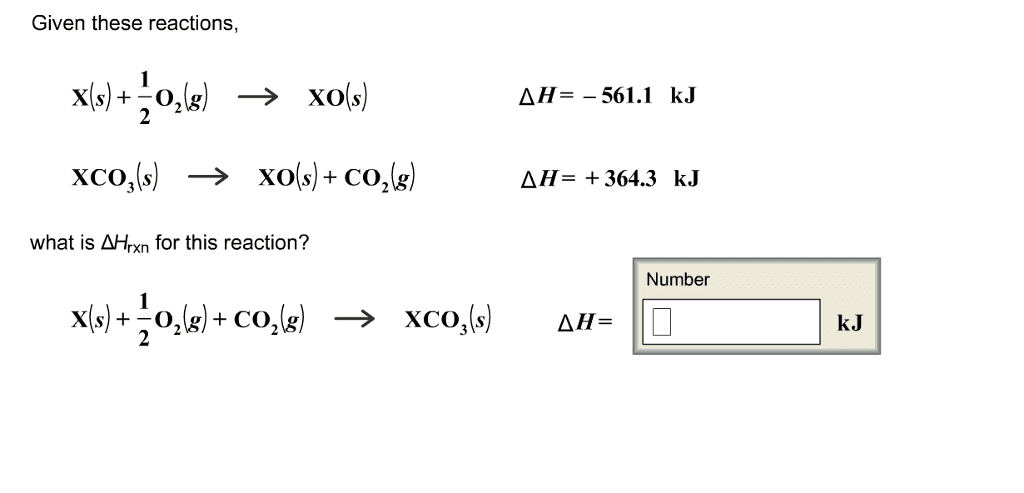
Given these reactions, X(s) + 1/2 O_2 (g) rightarrow XO(s) XCO_3(s) rightarrow XO(s) + CO_2(g) what is delta H_rxn for this reaction? delta H= -561.1 kJ delta H= +364.3 kJ X(s) + 1/2 O_2(g) rightarrow XCO_3(s) Delta H =

Given these reactions, X(s) + 1/2 O_2 (g) rightarrow XO(s) XCO_3(s) rightarrow XO(s) + CO_2(g) what is delta H_rxn for this reaction? delta H= -561.1 kJ delta H= +364.3 kJ X(s) + 1/2 O_2(g) rightarrow XCO_3(s) Delta H =
Jarrod RobelLv2
23 Oct 2019
