1
answer
0
watching
358
views
18 Nov 2019
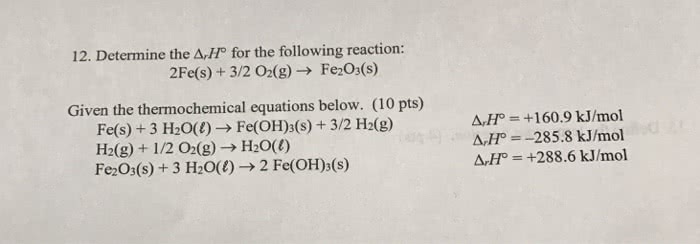
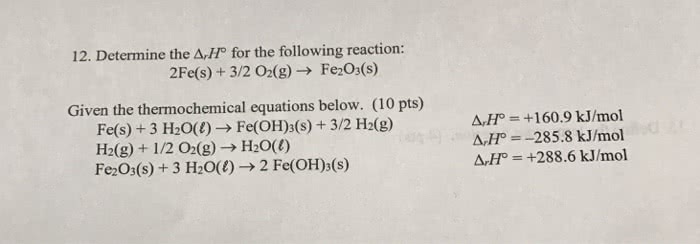
Determine the Delta_r H degree for the following reaction: 2Fe(s) + 3/2 O_2(g) rightarrow Fe_2O_3(s) Given the thermochemical equations below. Fe(s) + 3 H_2O(l) rightarrow Fe(OH)_3(s) + 3/2 H2(g) Delta_r H degree = +160.9 kJ/mol H2(g) rightarrow 1/2 O_2(g) rightarrow H_2O(l) Delta_r H degree = -285.8 kJ/mol Fe2O_3(s) rightarrow 3 H_2O(l) rightarrow 2 Fe(OH)_3(s) Delta_r H degree = +288.6 kJ/mol
Beverley SmithLv2
17 Feb 2019
