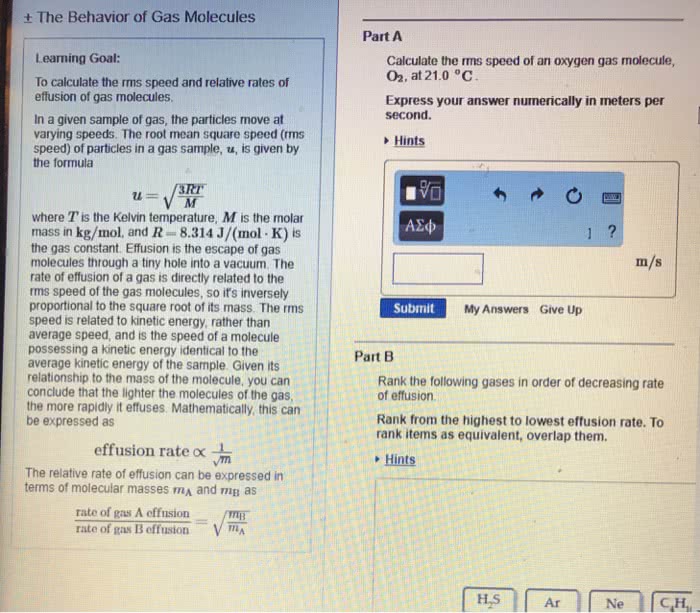
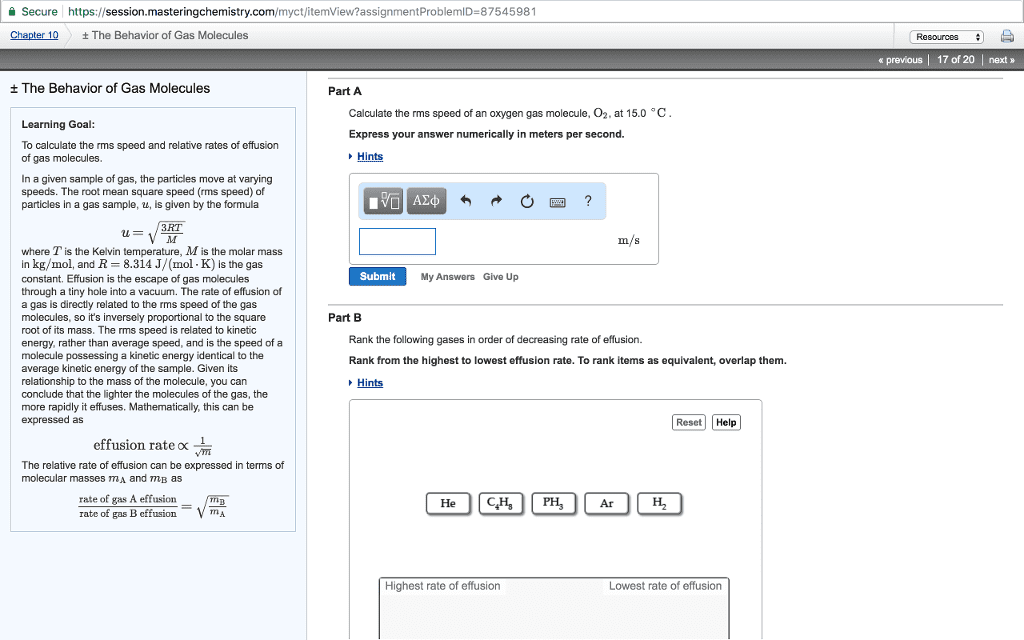
1.Effusion is a process in which a gas escapes from a container through a small opening into a vacuum. In a pair of experiments, a sample of tetrafluoroethylene, C2F4, effuses at a rate of 4.6 à 10-6 mol/h and an unknown gas effuses at a rate of 4.0Ã10-6 mol/h. Identify the unknown gas. 2.Molecules in a sample of a gas move at a variety of speeds. Molecular speed can be described by the root-mean-square speed of the gas, which is the square root of the average of the squares of the speeds of all the gas molecules. What is the rms speed of a sample of CO2 at 69.99 °C, in m/s? 3.Nitrogen monoxide is a pollutant commonly found in smokestack emissions. One way to remove it is to react it with ammonia. 4NH3(g) + 6NO(g) â 5N2(g) + 6H2O(â) How many liters of ammonia are required to change 41.3 L of nitrogen monoxide to nitrogen gas? Assume 100% yield and that all gases are measured at the same temperature and pressure. 4.A 1.78-g sample of an unknown gas has a volume of 884 mL and a pressure of 968 mmHg at 57.1 °C. Calculate the molar mass of this compound.
1.Effusion is a process in which a gas escapes from a container through a small opening into a vacuum. In a pair of experiments, a sample of tetrafluoroethylene, C2F4, effuses at a rate of 4.6 à 10-6 mol/h and an unknown gas effuses at a rate of 4.0Ã10-6 mol/h. Identify the unknown gas. 2.Molecules in a sample of a gas move at a variety of speeds. Molecular speed can be described by the root-mean-square speed of the gas, which is the square root of the average of the squares of the speeds of all the gas molecules. What is the rms speed of a sample of CO2 at 69.99 °C, in m/s? 3.Nitrogen monoxide is a pollutant commonly found in smokestack emissions. One way to remove it is to react it with ammonia. 4NH3(g) + 6NO(g) â 5N2(g) + 6H2O(â) How many liters of ammonia are required to change 41.3 L of nitrogen monoxide to nitrogen gas? Assume 100% yield and that all gases are measured at the same temperature and pressure. 4.A 1.78-g sample of an unknown gas has a volume of 884 mL and a pressure of 968 mmHg at 57.1 °C. Calculate the molar mass of this compound.