1
answer
0
watching
224
views
18 Nov 2019
This is all I'm given.
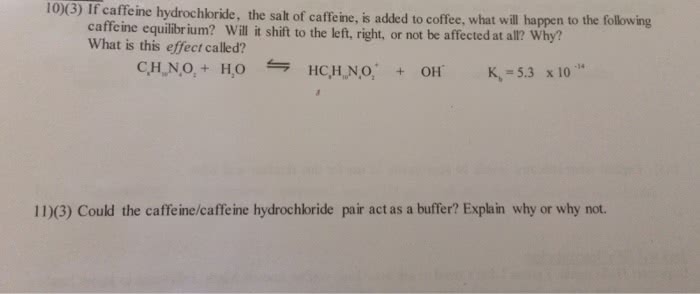
If caffeine hydrochloride. the salt of caffeine, is added to coffee. what will happen to the following caffeine equilibrium? Win it shift to the left, right, or not be affected at all? Why? What is this effect called? Could the caffeine/caffeine hydrochloride pair act as a buffer? Explain why or why not.
This is all I'm given.
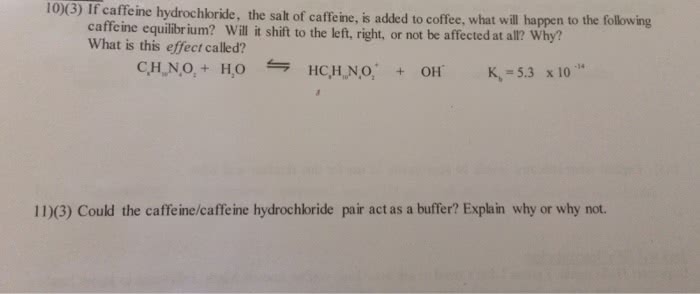
If caffeine hydrochloride. the salt of caffeine, is added to coffee. what will happen to the following caffeine equilibrium? Win it shift to the left, right, or not be affected at all? Why? What is this effect called? Could the caffeine/caffeine hydrochloride pair act as a buffer? Explain why or why not.
1
answer
0
watching
224
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jean KeelingLv2
23 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232



