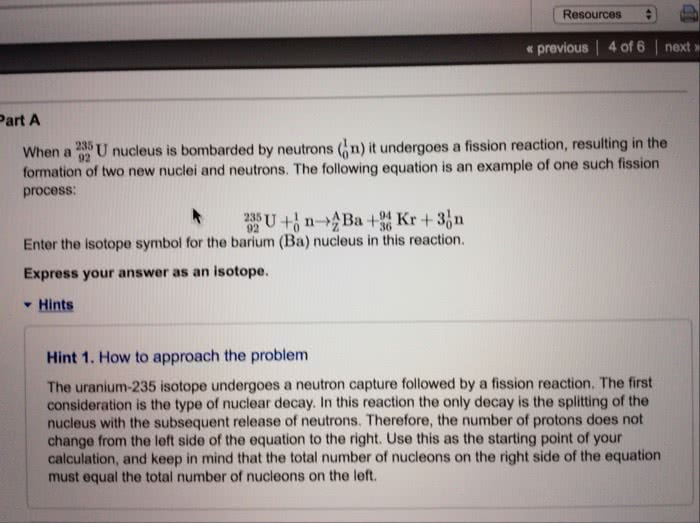
a.) The atomic mass unit is defined as 1/12th of the mass of the isotope (blank)-(blank) (use the first blank to give the element and the second to give the mass number) and is equivalent to (blank) Ã 10-27 kg.
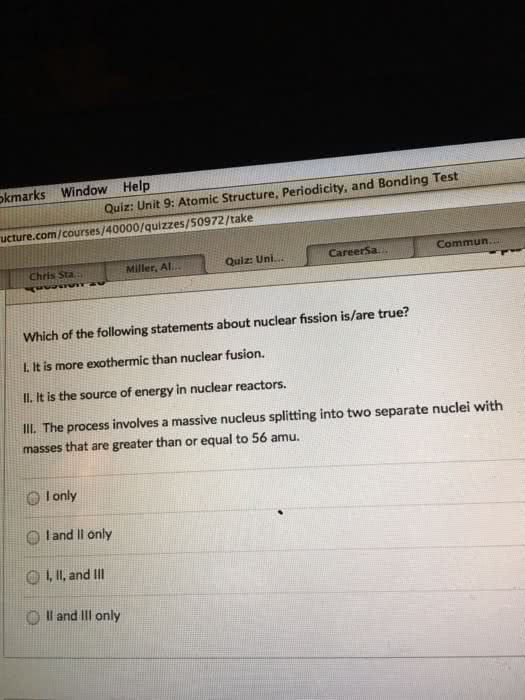
b.) Nuclear fission takes [ Select ] ["two or more small nuclei", "a large nucleus"] and [ Select ] ["merges it into a larger nucleus", "splits it into smaller nuclei"] ; nuclear fusion takes [ Select ] ["two or more smaller nuclei", "a larger nucleus"] and [ Select ] ["splits it into smaller nuclei", "merges them into a larger nucleus"] . Both processes release energy because the products have [ Select ] ["greater", "less"] binding energy per nucleon than the starting nuclei. However, at some point it will take energy to split or fuse a nucleus; this happens for a mass number of about [ Select ] ["56", "65", "235", "4"] .
a.) The atomic mass unit is defined as 1/12th of the mass of the isotope (blank)-(blank) (use the first blank to give the element and the second to give the mass number) and is equivalent to (blank) Ã 10-27 kg.
b.) Nuclear fission takes [ Select ] ["two or more small nuclei", "a large nucleus"] and [ Select ] ["merges it into a larger nucleus", "splits it into smaller nuclei"] ; nuclear fusion takes [ Select ] ["two or more smaller nuclei", "a larger nucleus"] and [ Select ] ["splits it into smaller nuclei", "merges them into a larger nucleus"] . Both processes release energy because the products have [ Select ] ["greater", "less"] binding energy per nucleon than the starting nuclei. However, at some point it will take energy to split or fuse a nucleus; this happens for a mass number of about [ Select ] ["56", "65", "235", "4"] .