A particular sample of vinegar has a pH of 3.00.
If acetic acid is the only acid that vinegar contains (Ka=1.8Ã10â5), calculate the initial concentration of acetic acid in the vinegar.
Express your answer using two significant figures.
A particular sample of vinegar has a pH of 3.00.
If acetic acid is the only acid that vinegar contains (Ka=1.8Ã10â5), calculate the initial concentration of acetic acid in the vinegar.
Express your answer using two significant figures.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
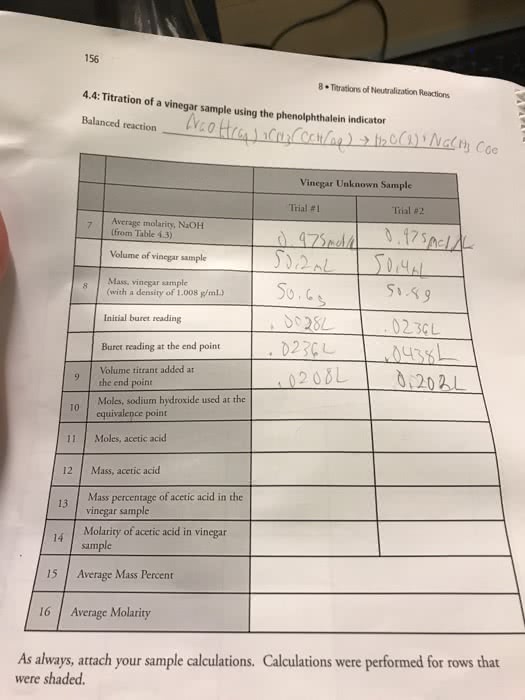
Historically, vinegar played an important role in the preservation of food because of the acidity and antimicrobial properties of its main component, acetic acid. There are many types of vinegar found at your local grocery store, including balsamic, rice, red wine, and apple cider vinegars. A titration will help you determine the concentration of acetic acid (CH3COOH) in vinegar. The balanced equation for the titration of acetic acid in vinegar with sodium hydroxide (NaOH) is as follows.
CH3COOH(aq) + NaOH(aq) â H2O(l) + NaCH3COO(aq)
Assume you titrated a sample of vinegar using 0.145 M of sodium hydroxide. The results of this titration are shown in the following table.
| Volume of Vinegar Titrated (mL) | 5.00 |
|---|---|
| Initial Volume of NaOH (mL) | 0.35 |
| Final Volume of NaOH (mL) | 29.50 |
(a) Use the data in the above table to determine the vinegar's acetic acid concentration in moles per liter.
0.845 mol/L
(b) Calculate the percent acetic acid in the titrated vinegar. Note that 1% = (1 g acetic acid/100 mL vinegar) Ã 100%.
_____ %
Please find and show work for part B