1
answer
0
watching
332
views
18 Nov 2019
which of the following pairs of substances could be used to construct a single redox electrode
which of the following pairs of substances could be used to construct a single redox electrode

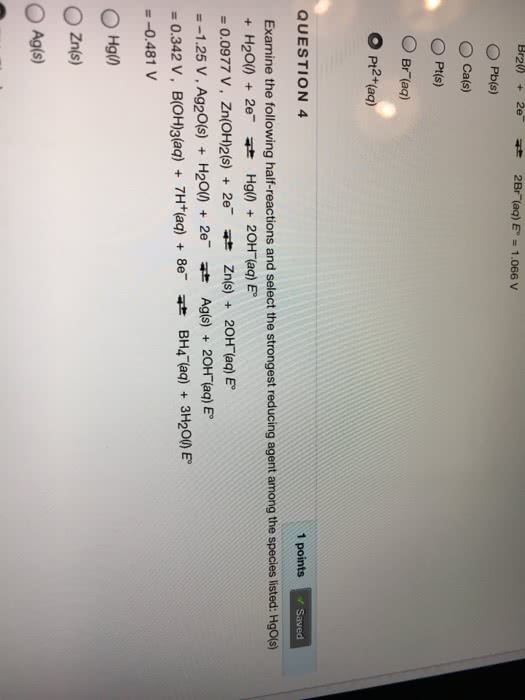
O Cals) ãPt(s) Br (aq) O Pi2+(aq) 1 points Savecd QUESTION 4 Saved Examine the following half-reactions and select the strongest reducing agent among the species listed: Hgos) + H2O) 2 Hg) 20H (aq) E = 0.0977 V, Zn(OH)2(s) + 2e- Zn(s) + 20H-(aq)E" =-1.25V, Ag2O(s) + H20() + 2e- Ag(s) + 20H-(aq)E" = 0.342 V, B(OH)3(aq) + 7H+(aq) + 8e- zt BH4-(aq) + 3H2onE" =-0.481 V O Zn/s) OAgis)
Sixta KovacekLv2
26 Apr 2019
