1
answer
0
watching
100
views
18 Nov 2019




S:= k In W V-J/C LAmp ICoulomb/sec R=8.314 J/Mol K (20 points) For the electrochemical cell Cr(s) Cr3* (aq) I| Br2 (I) | Br (aq) | Pt (s) a) Draw the cell and label anode, cathode, salt bridge, show the direction of electrons and write the net ionic equation. b) Calculate the standard cell potential.
1
answer
0
watching
100
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Keith LeannonLv2
9 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
| Based on the following electrochemical cell, what is the standard reduction potential of metal M at 298 K? (R = 8.314 J/K ⢠mol, F = 96500 C/mol) |
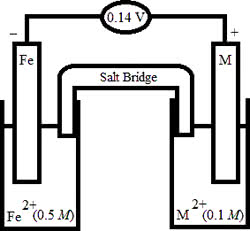 |
| Half-Reaction | E° (V) |
| Fe2+(aq) + 2eâ â Fe(s) | â0.44 |
A) â0.54 V
B) +0.60 V
C) â0.30 V
D) +0.56 V
E) â0.28 V
0.14 V Fe Salt Bridge (0.33 2+ M (0.1M)


