1
answer
0
watching
1,618
views
17 Nov 2019
Which of the following reactions will occur spontaneously as written?
3Fe (s) + 2Cr3+ (aq) ? 2Cr (s) + 3Fe2+ (aq) Sn4+ (aq) + Fe2+ (aq) ? Sn2+ (aq) + Fe (s) Sn4+ (aq) + Fe3+ (aq) ? Sn2+ (aq) + Fe2+ (aq) 3Sn4+ (aq) + 2Cr (s) ? 2Cr3+ (aq) + 3Sn2+ (aq) 3Fe2+ (aq) ? Fe (s) + 2Fe3+ (aq)
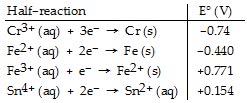
Half-reaction Cr+ (aq) 3e (s) 0.74 Fe2+ (aq) 2e Fe (s) -0.440 Fe3+ (aq) e Fe2 (s) +0.771 Snt+ (aq) 2e- Sn2+ (aq) l +0.154
Which of the following reactions will occur spontaneously as written?
| 3Fe (s) + 2Cr3+ (aq) ? 2Cr (s) + 3Fe2+ (aq) |
| Sn4+ (aq) + Fe2+ (aq) ? Sn2+ (aq) + Fe (s) |
| Sn4+ (aq) + Fe3+ (aq) ? Sn2+ (aq) + Fe2+ (aq) |
| 3Sn4+ (aq) + 2Cr (s) ? 2Cr3+ (aq) + 3Sn2+ (aq) |
| 3Fe2+ (aq) ? Fe (s) + 2Fe3+ (aq) |
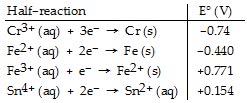
Half-reaction Cr+ (aq) 3e (s) 0.74 Fe2+ (aq) 2e Fe (s) -0.440 Fe3+ (aq) e Fe2 (s) +0.771 Snt+ (aq) 2e- Sn2+ (aq) l +0.154
Nestor RutherfordLv2
4 Feb 2019
