1
answer
0
watching
94
views
17 Nov 2019
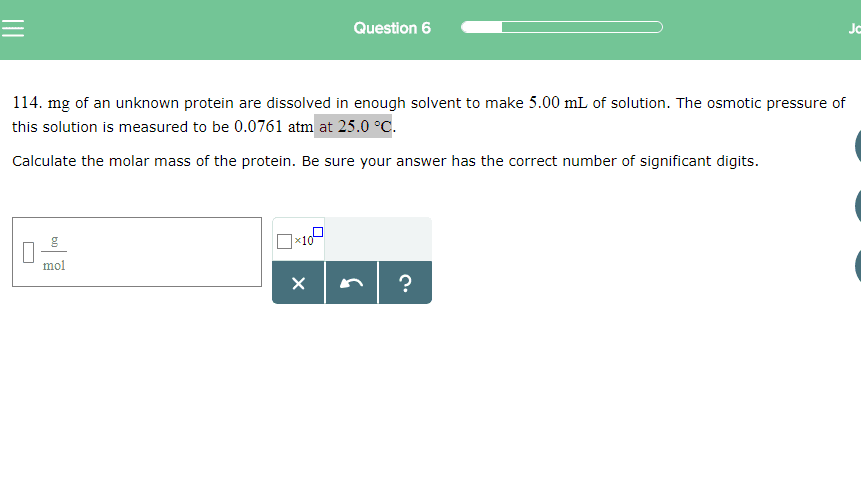
An 18.6 mg sample of a protein was dissolved in water to produce 5.00mL of solution at 25 degrees celsius. The osmotic pressure of this solution was measured and found to be 2.42 x10-3atm. What is the molar mass of this protein?
An 18.6 mg sample of a protein was dissolved in water to produce 5.00mL of solution at 25 degrees celsius. The osmotic pressure of this solution was measured and found to be 2.42 x10-3atm. What is the molar mass of this protein?
Beverley SmithLv2
3 Sep 2019