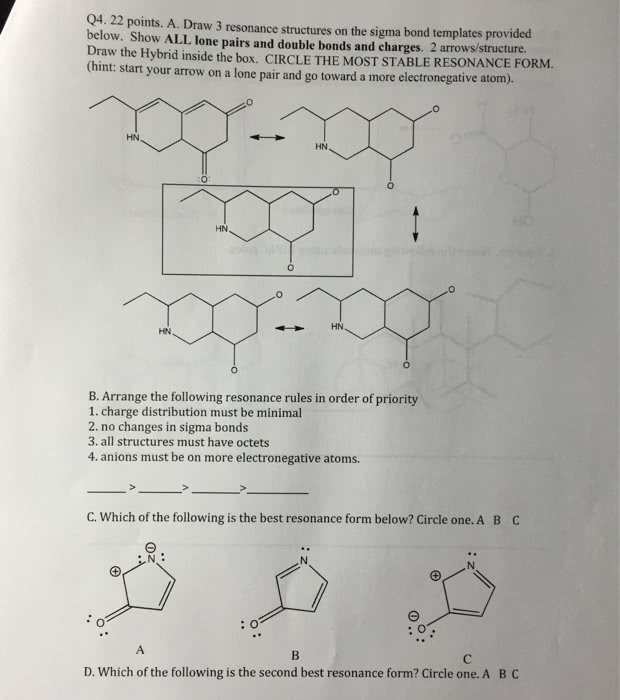
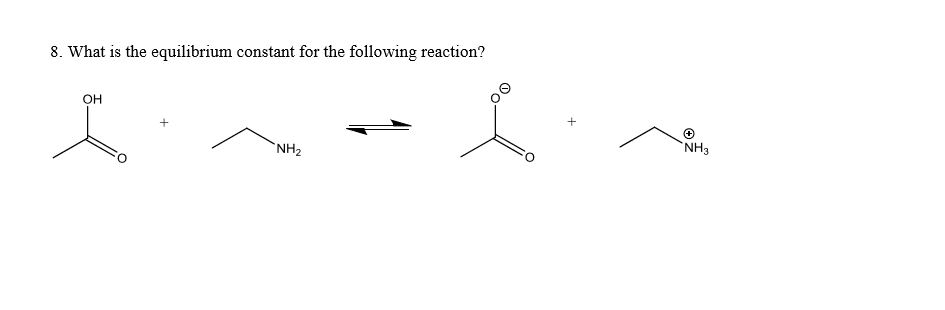
See the figure below, The resonance forms show are not correct because
a.) more resonance forms do not necessarily mean greater stabilization
b.) the second resonance form hasincomplete octets
c.)resonance form can only involve delocalization of pi and sigma electrons
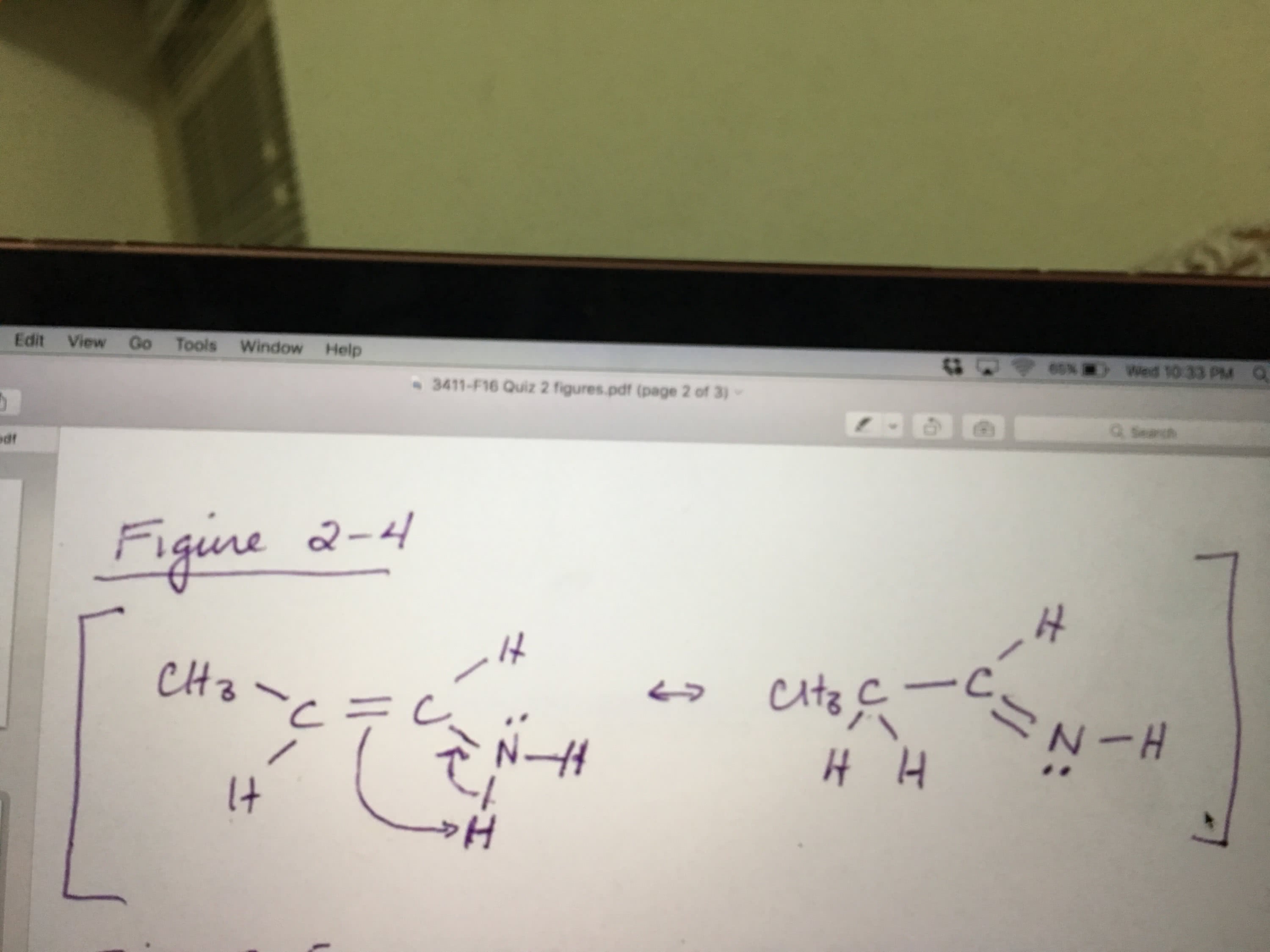
In the figure below, which resonance form is likely to be the least contributor to the actual structure.
a.) 1
b.) 2
c.) 3
d.) 1 & 2
e.) 2 & 3

The best reasoning for the answer in the above question is ?
a.) Identical structures are equally important
b.) Complete octets are more important
c.) Minimizing formal charges is more important
d.)Having positive formal charges on electropositive atoms and negative formal charges on electronegative atoms is important.
1.) Both A & B
2.) Both A & D
3.) Both B & C
4.) Both B & D
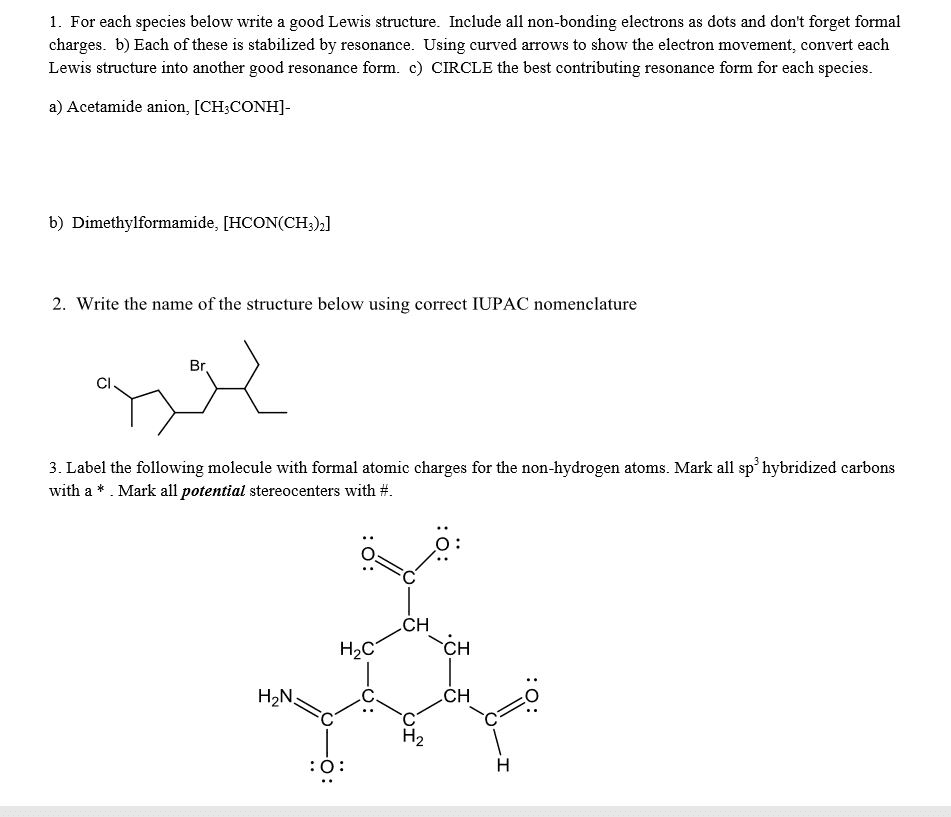
See the figure below, The resonance forms show are not correct because
a.) more resonance forms do not necessarily mean greater stabilization
b.) the second resonance form hasincomplete octets
c.)resonance form can only involve delocalization of pi and sigma electrons

In the figure below, which resonance form is likely to be the least contributor to the actual structure.
a.) 1
b.) 2
c.) 3
d.) 1 & 2
e.) 2 & 3

The best reasoning for the answer in the above question is ?
a.) Identical structures are equally important
b.) Complete octets are more important
c.) Minimizing formal charges is more important
d.)Having positive formal charges on electropositive atoms and negative formal charges on electronegative atoms is important.
1.) Both A & B
2.) Both A & D
3.) Both B & C
4.) Both B & D