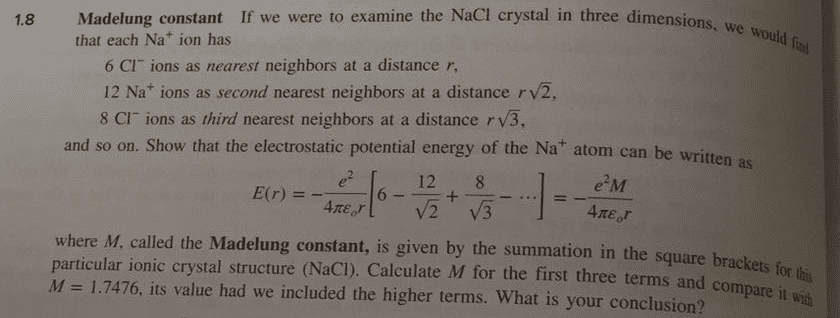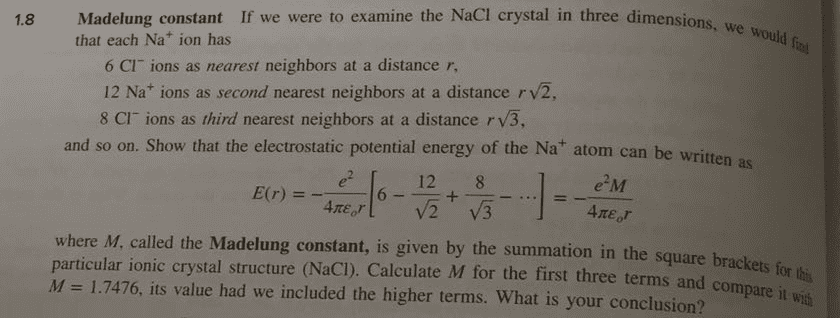1
answer
0
watching
124
views
17 Nov 2019
In most instances in metal systems, we consider only the first or second near-neighbor interactions; however, in ionic crystals the lattice energy depends upon interactions among all the ions in the system. Consider a Na+ ion in the NaCl-structure which has
(a) 6 Cl ions at a distance of r; (b) 2d near-neighbors are 12 Na+ at Ã2r; (c) 3d n-n are 8 Cl- at Ã3r; (d) 4th n-n are 6 Na+ at 2r; (e) 5th n-n are 24 Cl- at Ã5r.
i) Write the first three terms for determining lattice energy; ii) the first four terms; iii) the first five terms. Compare with the value of the Madelung constant (1.7475). What is the significance of a negative value? What does the calculations tell you about binding in an ionic crystal?
In most instances in metal systems, we consider only the first or second near-neighbor interactions; however, in ionic crystals the lattice energy depends upon interactions among all the ions in the system. Consider a Na+ ion in the NaCl-structure which has
(a) 6 Cl ions at a distance of r; (b) 2d near-neighbors are 12 Na+ at Ã2r; (c) 3d n-n are 8 Cl- at Ã3r; (d) 4th n-n are 6 Na+ at 2r; (e) 5th n-n are 24 Cl- at Ã5r.
i) Write the first three terms for determining lattice energy; ii) the first four terms; iii) the first five terms. Compare with the value of the Madelung constant (1.7475). What is the significance of a negative value? What does the calculations tell you about binding in an ionic crystal?
Elin HesselLv2
4 Jan 2019