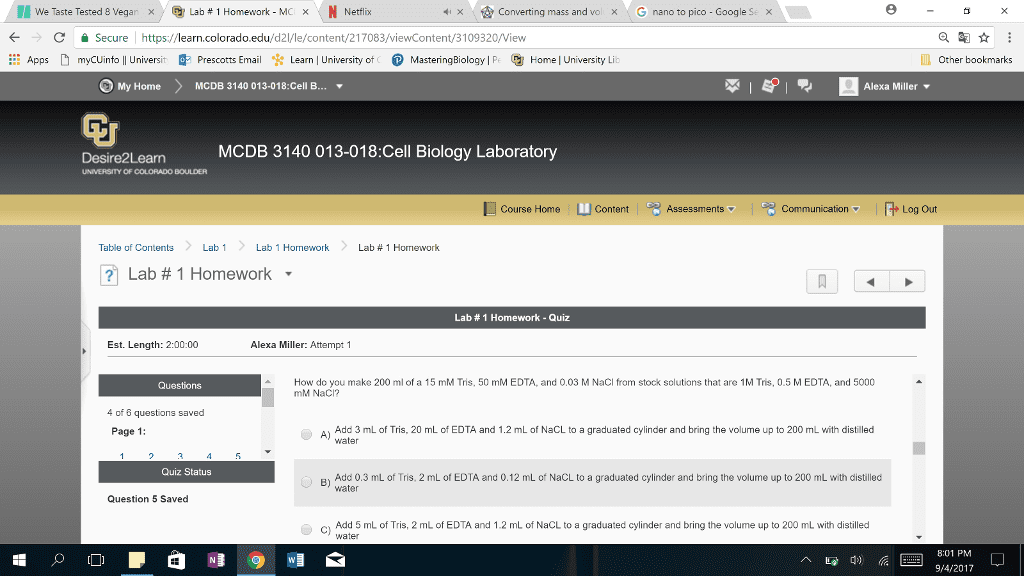
These are all questions I got wrong on a quiz. Please help explain to me what the correct answers are and why. Thank you!
1.) You have a solution that contains 80mM Calcium carbonate. You need to make 50mls of a solution that contains 4mM calcium carbonate. How many mls of the 80mM solution do you need to use?
A.) 2.25mls
B.) 25mls
C.) 2.5mls
D.) 8mls
E.) 47.75mls
F.) .8mls
2.) In order to make 100mls a 6.5mM solution of magnesium sulfate what do you need to know?
A.) the molecular weight of magnesium sulfate
B.) the solvent which you are making it in
C.) the molarity of water
3.) You have a stock solution of 10M NaCl. You want to make 100mls of a 1M NaCl solution. How would you dilute the stock to create this solution?
A.) add 10mls of 10M NaCl to 90mls of water
B.) add 1 ml of 10M NaCl to 99mls of water
C.) add 10 mls of 10M NaCL to 100mls of water
D.) add 5mls of 10M NaCl to 95mls of water
4.) A chemist wants to prepare a stock solution of H2SO4 so that samples of 20.00 mL will produce a solution with a concentration of 0.50 M when added to 80.0 mL of water. What should the molarity of the stock solution be?
These are all questions I got wrong on a quiz. Please help explain to me what the correct answers are and why. Thank you!
1.) You have a solution that contains 80mM Calcium carbonate. You need to make 50mls of a solution that contains 4mM calcium carbonate. How many mls of the 80mM solution do you need to use?
A.) 2.25mls
B.) 25mls
C.) 2.5mls
D.) 8mls
E.) 47.75mls
F.) .8mls
2.) In order to make 100mls a 6.5mM solution of magnesium sulfate what do you need to know?
A.) the molecular weight of magnesium sulfate
B.) the solvent which you are making it in
C.) the molarity of water
3.) You have a stock solution of 10M NaCl. You want to make 100mls of a 1M NaCl solution. How would you dilute the stock to create this solution?
A.) add 10mls of 10M NaCl to 90mls of water
B.) add 1 ml of 10M NaCl to 99mls of water
C.) add 10 mls of 10M NaCL to 100mls of water
D.) add 5mls of 10M NaCl to 95mls of water
4.) A chemist wants to prepare a stock solution of H2SO4 so that samples of 20.00 mL will produce a solution with a concentration of 0.50 M when added to 80.0 mL of water. What should the molarity of the stock solution be?