1
answer
1
watching
1,573
views
17 Nov 2019
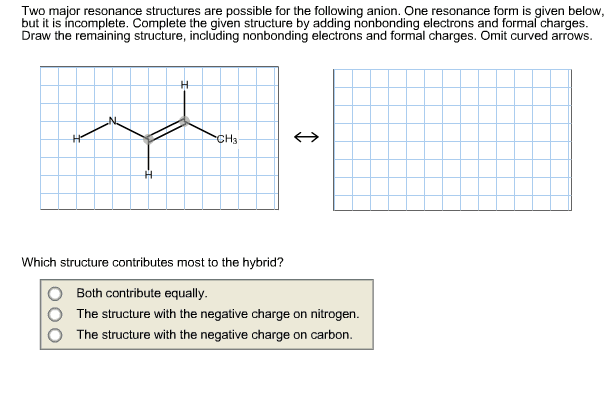
1.Draw one valid Lewis structure (including all lone pair electrons and any formal charges) for the anion
(CH2CN)â.
Assume the atoms are arranged as shown below.
H C C N H
2. Follow the curved arrows to draw a second resonance structure for the following species.
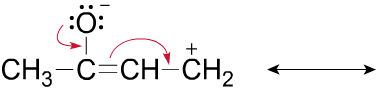
1.Draw one valid Lewis structure (including all lone pair electrons and any formal charges) for the anion
(CH2CN)â.
Assume the atoms are arranged as shown below.
| H | C | C | N |
| H |
2. Follow the curved arrows to draw a second resonance structure for the following species.
Beverley SmithLv2
22 Aug 2019