
Please help, I have the answer but don't know how to do the problem completely.
Thanks in advance.
16. How much energy (in Joules) is carried by one photon of violet light with λ= 400 nm? Answer: 4.96 x10-19 J

Please help, I have the answer but don't know how to do the problem completely.
Thanks in advance.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
We are supposed to calculate (approximatly) what n level each hydrogen emission start at. We know they all end at 2. This is what I have got so far but I am stuck on how to apply the calculations for the n level. Help!
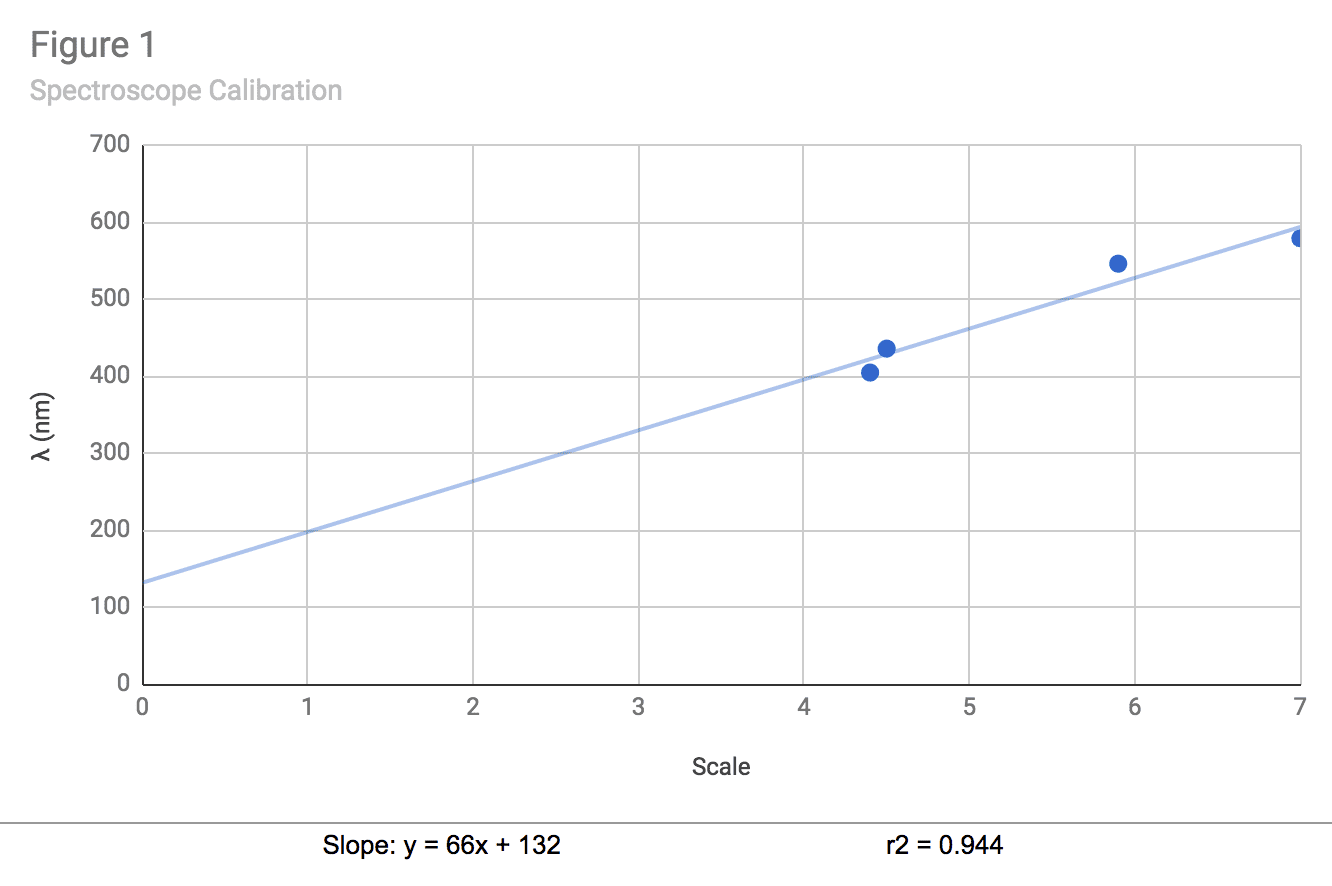
| Table 2: Spectroscope Calibration | |
| Hg λ | Scale |
| Violet: 404.7 | 4.4 |
| Blue: 435.8 | 4.5 |
| Green: 546.1 | 5.9 |
| Yellow: 579.0 | 7.0 |
| Table 3: H spectra | |||
| Color | Scale (x) | λ (nm) | n-level |
| Violet | 4.2 | 409nm | |
| Blue | 4.4 | 422nm | |
| Green | 4.9 | 455nm | |
| Red | 5.8 | 515nm | |
| Calculations | ||||
| Energy of N levels | Wavelength using calibration curve | E | Equation 2 | n-level |
| -2.18 x 10^-18J (1/3²) = -2.42 x 10^-19 J | Violet: 66(4.2) + 132 = 409nm | (6.63 x 10â»Â³â´)(3 x 10â¸)409 x 10â»â¹= 4.86 x 10^-19 | -2.18 x 10^-18J[(12²) - (13²)] = -3.03 x 10^-19 | |
| -2.18 x 10^-18 (1/4²) = -1.36 x 10-19 J | Blue: 66(4.5) + 132 = 422nm | (6.63 x 10â»Â³â´)(3 x 10â¸)422 x 10â»â¹= 4.71 x 10^-19 | -2.18 x 10^-18J[(12²) - (14²)] = -5.68 x 10^-20 | |
| -2.18 x 10^-18 (1/5²) = -8.72 x 10^-20 J | Green: 66(4.9) + 132 = 455nm | (6.63 x 10â»Â³â´)(3 x 10â¸)455 x 10â»â¹= 4.37 x 10-19 | -2.18 x 10^-18J[(12²) - (15²)] = -1.19 x 10^-20 | |
| -2.18 x 10^-18(1/6²) = -6.06 x 10^-20 J | Red: 66(5.8) + 132 = 515nm | (6.63 x 10â»Â³â´)(3 x 10â¸)515 x 10â»â¹= 3.86 x 10^-19 | -2.18 x 10^-18J[(12²) - (16²)] = -2.65 x10^-21 | |

