1
answer
0
watching
3,667
views
28 Sep 2019
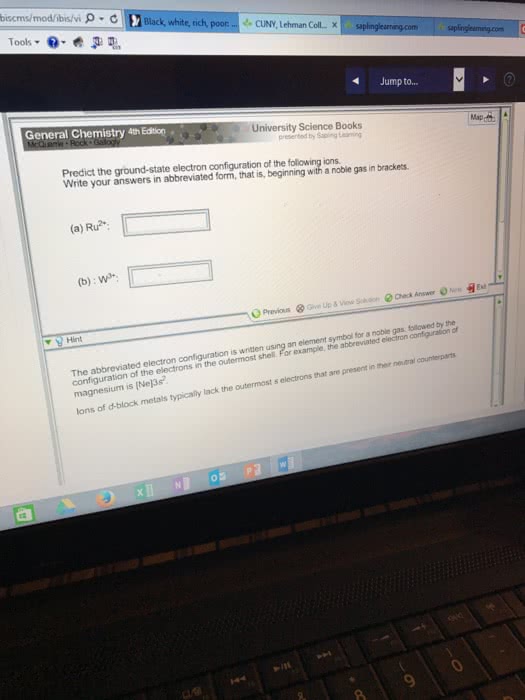
A: How many unpaired electrons are there in the predicted ground state configuration of a neutral ruthenium (Ru) atom according to the Aufbau principal?
B: What would be the predicted electron configuration for a Ni2+ ion?
C: How many unpaired electrons are there in the observed ground state configuration of a neutral ruthenium (Ru) atom?
D: Which group (column) of elements in the d-block has a different ground state distribution of electrons in the outermost s and d-orbitals for neutral atoms of each of its three elements?
E: Which elements in the d-block do NOT have the predicted electron configuration for the ground state of a neutral atom according to the Aufbau principal?
A: How many unpaired electrons are there in the predicted ground state configuration of a neutral ruthenium (Ru) atom according to the Aufbau principal?
B: What would be the predicted electron configuration for a Ni2+ ion?
C: How many unpaired electrons are there in the observed ground state configuration of a neutral ruthenium (Ru) atom?
D: Which group (column) of elements in the d-block has a different ground state distribution of electrons in the outermost s and d-orbitals for neutral atoms of each of its three elements?
E: Which elements in the d-block do NOT have the predicted electron configuration for the ground state of a neutral atom according to the Aufbau principal?
Deanna HettingerLv2
28 Sep 2019