1
answer
0
watching
188
views
16 Nov 2019
Balance the following reactions, assume acidic conditions
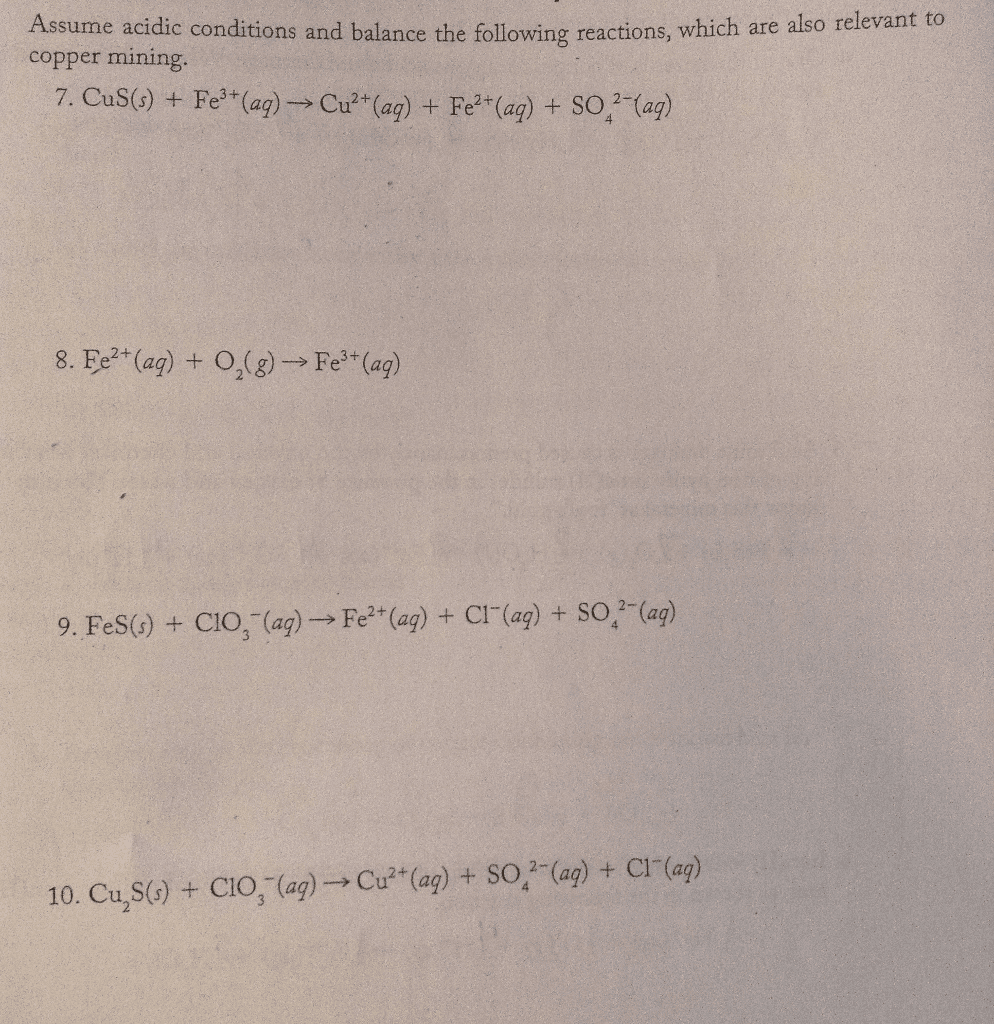
Assume acidic conditions and balance the following reactions, which are also relevant to copper mining. CuSO) + Fe^3+(aq) rightarrow Cu^2+ (aq) + Fe^2+(aq) + SO_4^2-(aq) Fe^2+(aq) + O_2(g) rightarrow Fe^3+(aq) FeS(s) + ClO_3 -(aq) rightarrow Fe^2+(aq) + Cl-(aq) + SO_4^2-(aq) Cu_2S(s) + CIO_3 -(aq) rightarrow -Cu^2+(aq) + SO_4^2-(aq) Cl^-(a q)
Balance the following reactions, assume acidic conditions
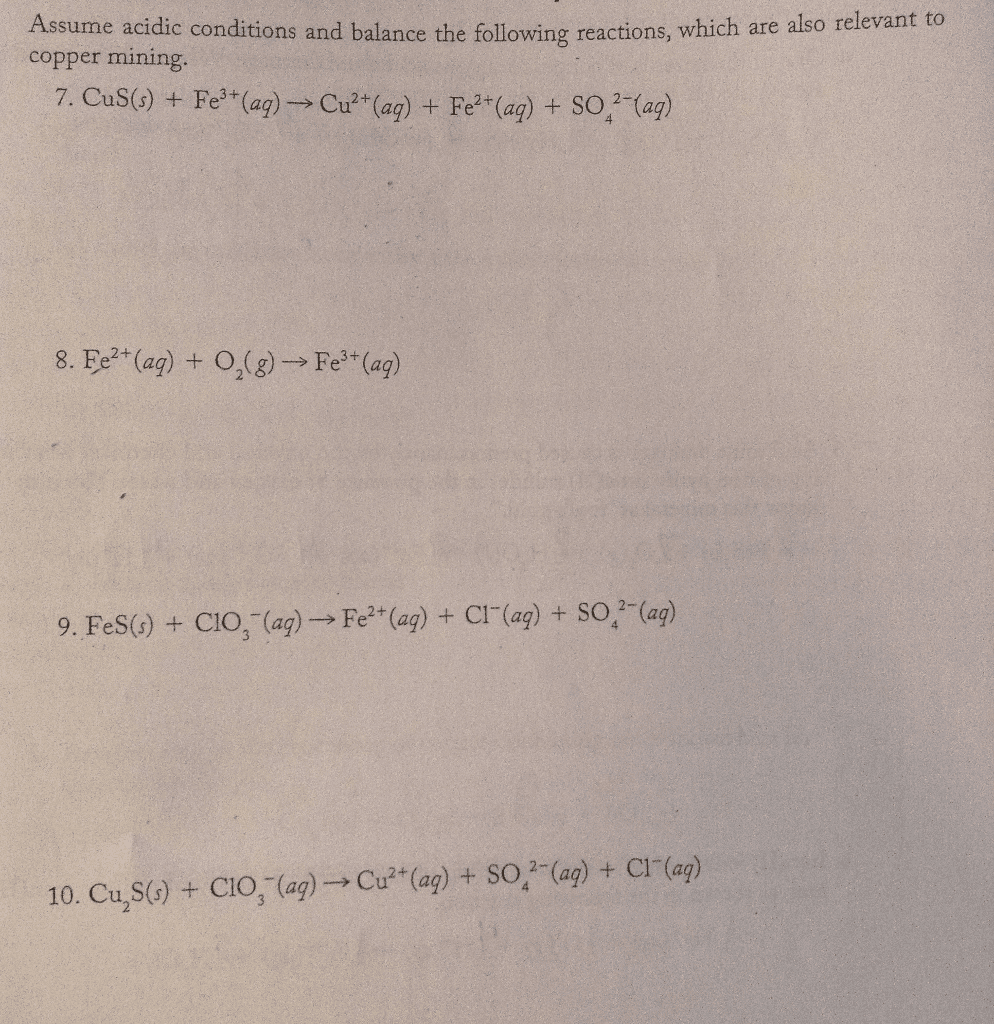
Assume acidic conditions and balance the following reactions, which are also relevant to copper mining. CuSO) + Fe^3+(aq) rightarrow Cu^2+ (aq) + Fe^2+(aq) + SO_4^2-(aq) Fe^2+(aq) + O_2(g) rightarrow Fe^3+(aq) FeS(s) + ClO_3 -(aq) rightarrow Fe^2+(aq) + Cl-(aq) + SO_4^2-(aq) Cu_2S(s) + CIO_3 -(aq) rightarrow -Cu^2+(aq) + SO_4^2-(aq) Cl^-(a q)
Sixta KovacekLv2
10 Mar 2019
