0
answers
0
watching
99
views
15 Nov 2019


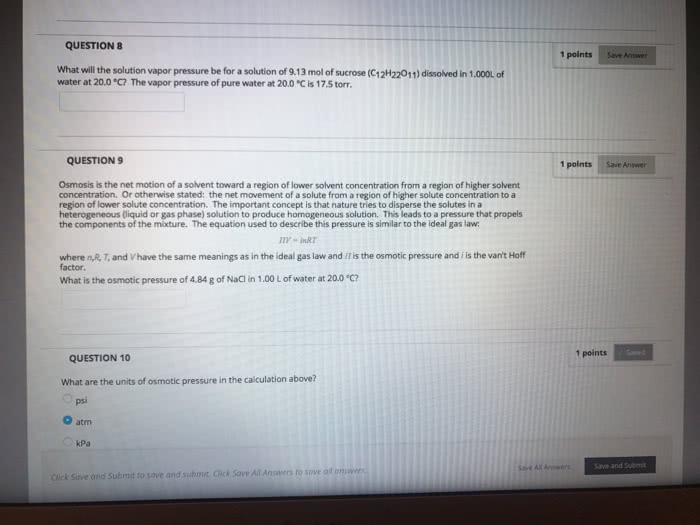
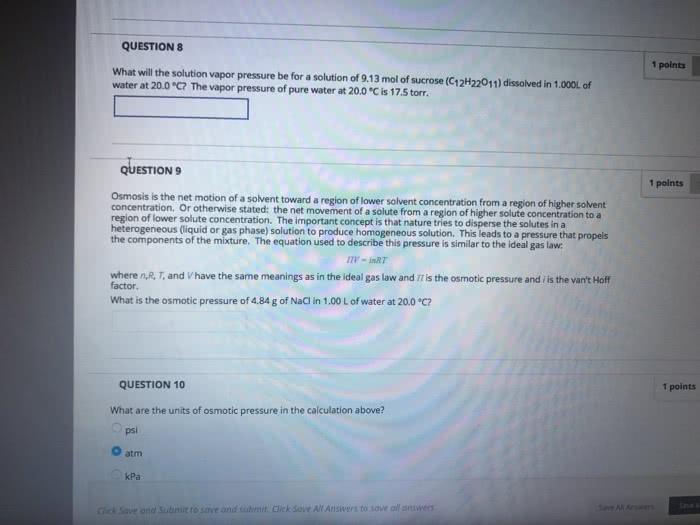
Use the Clausius Clapeyron equation and Raouit's law to find and ex- pression for the vapor pressure of a liquid solvent in a non-volatile so- lute+liquid solvent mixture with the solute concentration expressed in molals. Use your expression to calculate the vapor pressure of water for an ideal .200 molal solution of sucrose at 25 °C if the heat of vaporization is 40.69 k.J/mol.
0
answers
0
watching
99
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232