1
answer
0
watching
738
views
15 Nov 2019
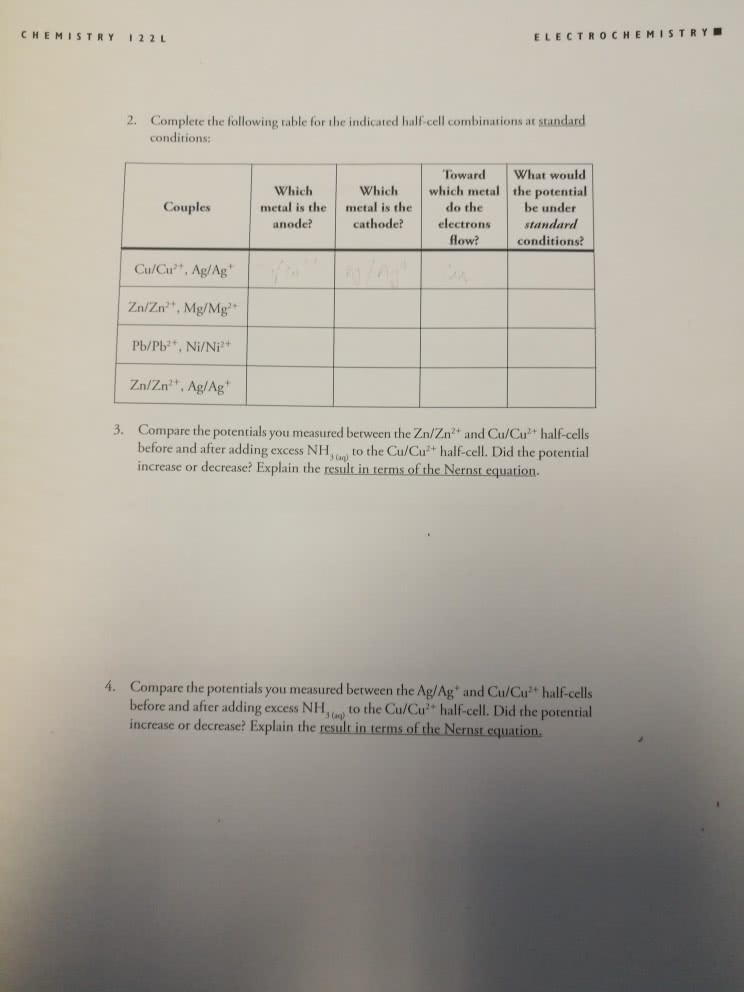
Write the balanced, net-ionic equations for the cell reactions for the following half-cell combinations at standard conditions. Be sure to consider direction of spontaneity.
A. Cu/Cu2+, Zn/Zn2+
B. Ag/Ag+, Mg/Mg2+
C. Zn/Zn2+, Pb/Pb2+
D. Ni/Ni2+, Ag/Ag+
E. Cu/Cu2+, Mg/Mg2+
Write the balanced, net-ionic equations for the cell reactions for the following half-cell combinations at standard conditions. Be sure to consider direction of spontaneity.
A. Cu/Cu2+, Zn/Zn2+
B. Ag/Ag+, Mg/Mg2+
C. Zn/Zn2+, Pb/Pb2+
D. Ni/Ni2+, Ag/Ag+
E. Cu/Cu2+, Mg/Mg2+
Reid WolffLv2
28 Aug 2019