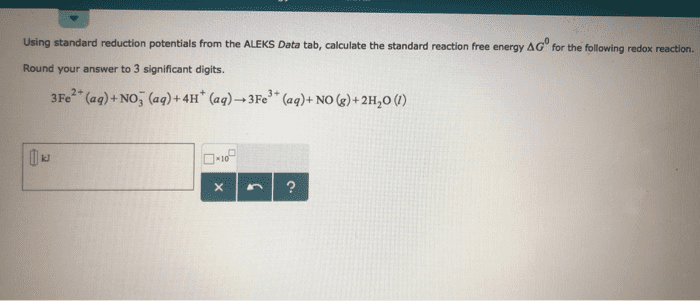
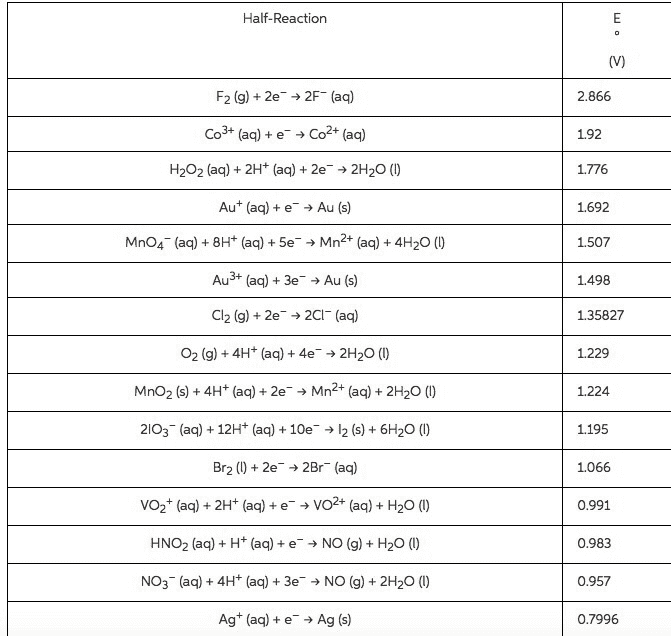
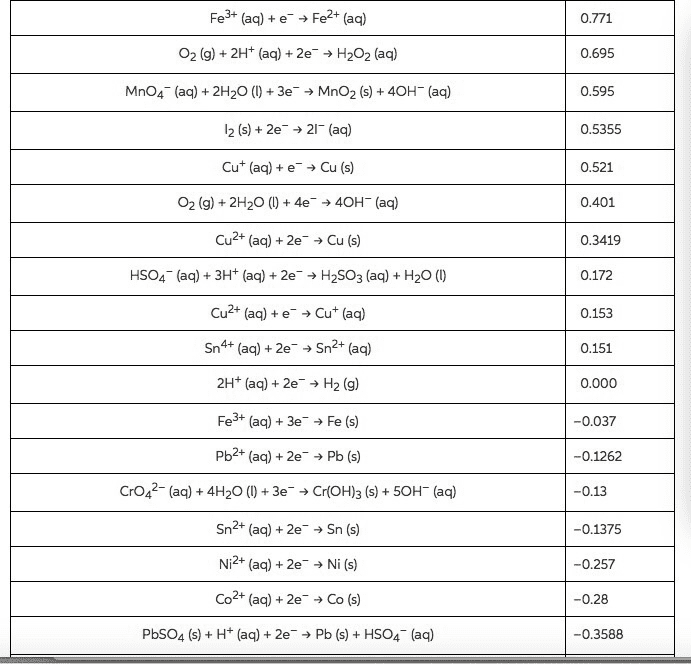
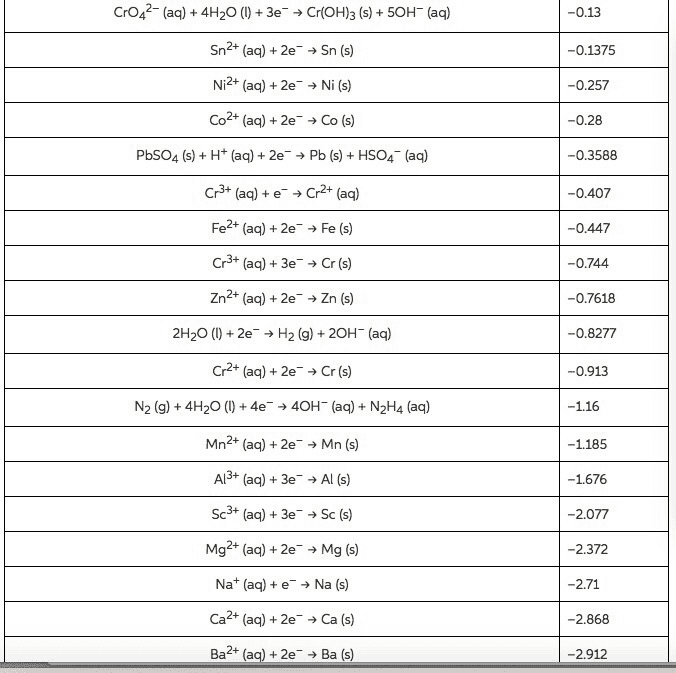
2
answers
5
watching
3,072
views
28 Sep 2019
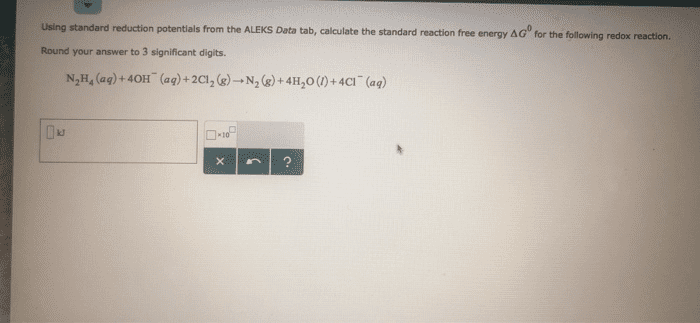
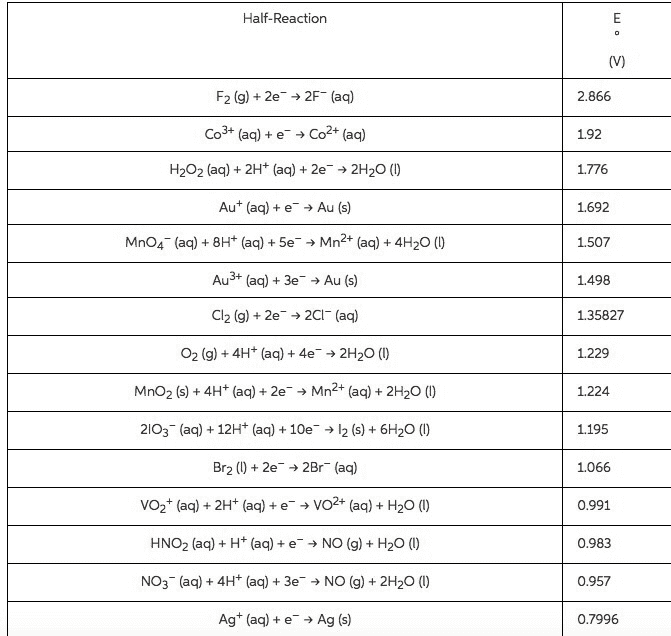
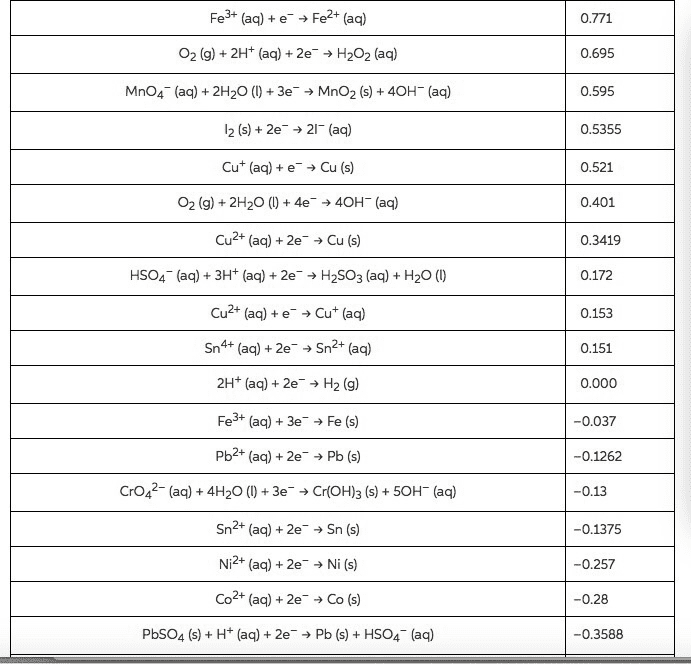
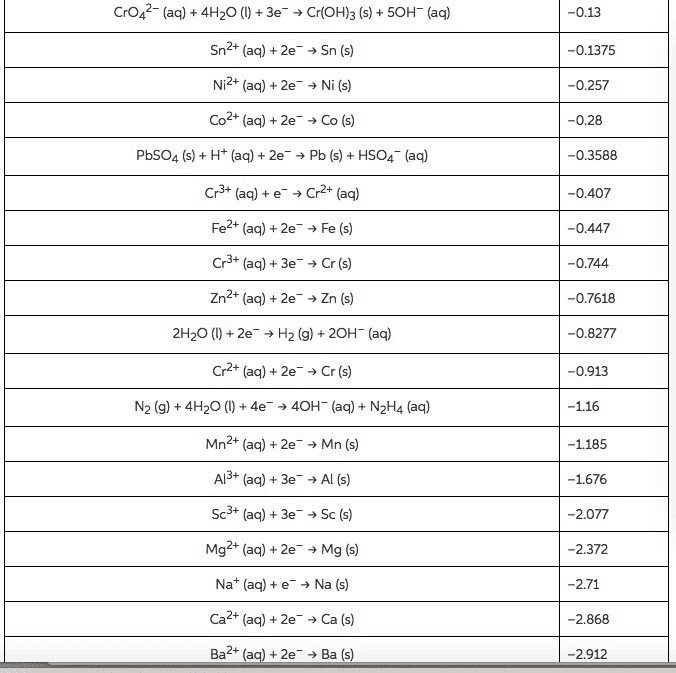
Balance the following equations in both acidic and basicenvironments (Show the half reactions right before they are addedtogether (after balancing all the components) and then show thebalanced equation):
1)Cr2O72-(aq) +C2H5OH(l) -->Cn3+(aq) + CO2(g)
2) Fe2+(aq) +MnO4-(aq) -->Fe3+(aq) +Mn2+(aq)
Balance the following equations in both acidic and basicenvironments (Show the half reactions right before they are addedtogether (after balancing all the components) and then show thebalanced equation):
1)Cr2O72-(aq) +C2H5OH(l) -->Cn3+(aq) + CO2(g)
2) Fe2+(aq) +MnO4-(aq) -->Fe3+(aq) +Mn2+(aq)
2
answers
5
watching
3,072
views
For unlimited access to Homework Help, a Homework+ subscription is required.
reallyvennyLv1
14 May 2023
Jarrod RobelLv2
28 Sep 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232